Emma Yasinski Scientific Writer Max Planck Florida Institute for Neuroscience
Researchers Are Developing Ways to Edit Some of the Most Difficult-to-Edit DNA-Neuronal DNA
Ryohei Yasuda, Ph.D., scientific director, and his team at the Max Planck Florida Institute of Neuroscience (MPFI) are working to understand the way individual cells in our brains change as we learn and form memories. One of their main goals is to understand how different proteins behave and impact the structure and function of an individual cell, but, much like the field of genetics was once limited by the inability to visualize the structure of DNA, their research has been limited by their ability to locate and visualize the many different types of proteins within a single cell. Current imaging methods do not provide contrast and specificity high enough to see distinct proteins. Plus, the best methods are time-consuming and expensive; it can take a year or more to develop engineered models.
Over the past few years, the development of CRISPR technology has helped scientists overcome countless genetic engineering challenges, and allowed them to edit genes with unmatched precision and speed, massively increasing clarity and cutting the cost of research requiring genetic engineering. The technique has been used in myriad ways to increase understanding and treatment of diseases and disorders, but some cells are more difficult to edit than others. Brain cells have proven especially difficult to manipulate using CRISPR.
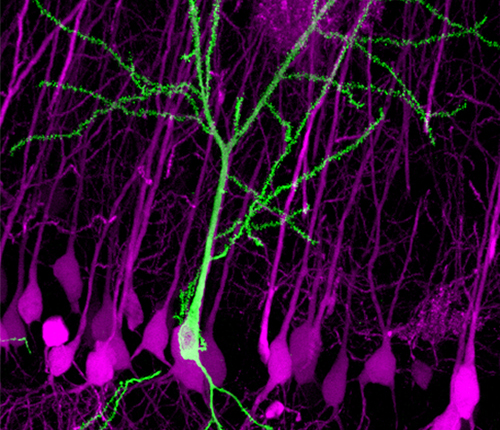
Recently, MPFI researchers Takayasu Mikuni, Ph.D., M.D., and Jun Nishiyama, Ph.D., M.D., and Dr. Yasuda were able to harness the power of the CRISPR/Cas9 system in order to create a quick, scalable, and high-resolution technique to edit neuronal DNA, which they called “SLENDR,” (single-cell labeling of endogenous proteins by CRISPR/Cas9-mediated homology-directed repair.) Using the technique, the researchers labeled several distinct proteins with fluorescence, and were able to observe protein localization in the brain that was previously invisible. That’s just the start of what researchers may be able to accomplish using this reliable, new technique for inserting genes into neurons.
CRISPR/Cas9 and Neurons
CRISPR is a tool built into bacterial DNA that the organisms use to fight infections. When a virus invades and attempts to insert its infectious DNA into that of a bacterial cell, a special section of the bacterial DNA, called CRISPR, cuts the viral DNA and renders it unable to wreak havoc on the bacteria. The organism then inserts a copy of the viral DNA into its own DNA to work as a type of adaptive immune system, to better recognize and defeat the invader in the future. As scientists have begun to understand how this system works, they have manipulated it to target and damage specific, functional genes in a variety of organisms, and in some cases, insert a new gene in its place.
Once the section of DNA is damaged, the technique relies on the cell to naturally repair its own DNA. There are two methods that the cell might use to accomplish this. One is homology-directed repair (HDR), the other is non-homologous end joining (NHEJ). HDR rebuilds or replaces the damaged locus of the genome, whereas NHEJ reattaches the damaged ends. When the reattachment occurs following the degradation of the ends, it often leads to the deletion of function of the gene (“knock-out” the gene). If a cell uses HDR to repair itself, scientists can include a desired gene in the CRISPR system that will be inserted into the DNA to replace the damaged gene.
Despite the impressive power of CRISPR system, its use in brain cells has been limited because by the time the brain has developed, its cells are no longer dividing. Most mature brain cells will repair themselves using NHEJ. The researcher can’t give the cell a gene to insert if it’s not going to insert one to begin with. While scientists can use CRISPR relatively easily to damage and knock out certain genes through NHEJ in the brain, the lack of cell division has made it very difficult for them to knock in desired sequences to genes, through HDR, with reliable precision. That’s where the SLENDR technique comes in.
SLENDR
SLENDR combines the power of the CRISPR/Cas9 system with the specificity and timing of in utero electroporation. Electroporation is a well-known technique used for introducing new material into cells and creating genetic knock-outs and knock-ins. Using in utero electroporation allows researchers to insert the CRISPR/CAS9 system into prenatal models, where brain cells are still developing and dividing. Thus, the broken DNA is still being repaired via HDR, giving researchers the opportunity to precisely modify a gene. This is a big deal. “I believe that SLENDR will be a standard tool for molecular and cellular neurobiology,” said Dr. Yasuda. “SLENDR provides a valuable means to determine subcellular localization of proteins, and will help researchers to determine the function of the proteins.”
In the recent study, the researchers at MPFI inserted a gene that made proteins of interest fluoresce under the microscope. They were even able to reliably label two different proteins with distinct colors at the same time in the same cell. The researchers were able to use the technique to visualize the proteins both in vivo and in vitro. And they were able to do it in a matter of days rather than years.
With existing knowledge of how brains develop, researchers can adjust the timing and position of the electroporation in utero to accurately target cells that will go on to populate particular cortical layers of the brain, even if they haven’t differentiated and moved to that layer yet.
The recent study used the technique primarily to tag certain proteins within brain cells and observe their behavior. But, with continued optimization, the method has the potential to elucidate immeasurable brain activities in both normal and diseased brains, and lead to a deeper understanding of brain function. “The most important part is that precise genome editing is possible in the brain. That’s what’s important,” said Dr. Nishiyama, post-doctoral researcher who worked on the study. “That’s the biggest thing.” Neuroscientists would be remiss to ignore its worth and not explore its potential.
Emma Yasinski is a scientific writer at Max Planck Florida Institute for Neuroscience. Correspondence should be directed to Ryohei Yasuda, Ph.D. ([email protected]), scientific director, Max Planck Florida Institute for Neuroscience.