Gunilla Enblad Science for Life Laboratory, Uppsala University
Hannah Karlsson Science for Life Laboratory, Uppsala University
Angelica S.I. Loskog Science for Life Laboratory, Uppsala University
The Role of Physical Barriers and Immunosuppression in Lymphoma
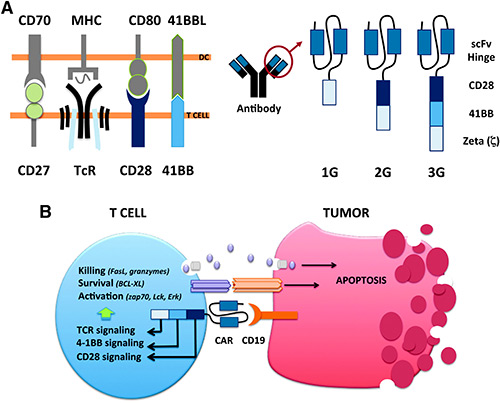
Chimeric antigen receptor (CAR) T-cells are T-cells genetically engineered to express a tumor-targeting receptor. The receptor is a chimera of a signaling domain of the T-cell receptor (TcR) complex and an antigen-recognizing domain, such as a single chain fragment (scFv) of an antibody.1 Hence, independently of the native TcR, CAR T-cells can recognize tumor cells via the CAR receptor. In contrast to TcR-mediated recognition of target cells via protein peptides displayed on major histocompatibility complex (MHC) molecules, the CAR is not dependent on MHC. The CAR molecule will recognize any target on the tumor cell surface and it is not limited to be a protein since antibodies can bind also carbohydrates and lipids. As for all targeted cancer therapeutics, the target needs to be specific for the cancer cells to avoid damage of healthy tissues. In many ways B-cell malignancy is the ideal indication for targeted therapy such as CAR T-cell therapy. B-cells are easily targeted via specific and selective markers such as CD19, CD20, and the Ig kappa or light chains. Considering that persisting problems with infectious disease because of B-cell deficiency can be handled with immunoglobulin replacement therapy, eradication also of the healthy B-cell population along with the malignant B-cells is manageable. Moreover, new B-cells will develop from the hematopoietic stem cells since these cells lack aforementioned B-cell markers and are, hence, not killed by CAR T-cells.
B-cell malignancy is a heterogeneous indication with both solid lesions and circulating cells in blood and bone marrow. Treatment of B-cell malignancy using CAR T-cells presents a unique opportunity to learn mechanisms of action of different CAR designs, to define on and off target toxicity, as well as to understand the limitations of CAR T-cells in terms of sensitivity to immune escape mechanisms and physical barriers of solid tumors.
To view the rest of this article click here.
Human Gene Therapy, published by Mary Ann Liebert, Inc., presents reports on the transfer and expression of genes in mammals, including humans. Human Gene Therapy and its companion publications can be viewed on the Human Gene Therapy website. The above article was first published the August 2015 issue of Human Gene Therapy. The views expressed here are those of the authors and are not necessarily those of Industrial Biotechnology, Mary Ann Liebert, Inc., publishers, or their affiliates. No endorsement of any entity or technology is implied.