Jim Zacka
Follow these instructions for healthy cultures and consistent results.
The process for isolating primary cells and preparing them for successful nonperfusion culture, while well understood, too often proves to be problematic. Here we review the basic methods and materials used and present an overall guide that, if followed, will lead to healthy cultures and successful procedures with the cells of choice.
It is important to note that there are many parameters that may affect the outcome of any particular primary cell isolation procedure including but not limited to tissue type, species, age, genetic modification (knockouts, etc.), enzyme type/quality, concentration, and incubation time and temperature.
Initial Processing
Tissue should be harvested and processed using sterile equipment, reagents and techniques, and appropriate personal protective equipment to avoid contamination. All enzymes and reagents can be sterile filtered through a 0.22 micron-pore-size membrane once reconstituted and fully solubilized.
Tissue is then generally minced or cut into two-to-four millimeter pieces with sterile scissors or a scalpel, and the pieces are added to the appropriate buffer, media or balanced salt solution on ice. After two-three washes to remove excess blood proteins the enzymes of choice (collagenase, neutral protease, papain, trypsin, etc.) are added, usually in an amount of between 0.5 and 1.5 mg/mL. The tissue is then incubated at optimum temperature (typically 37°C) for 30–90 minutes (or other time if indicated) and intermittently mixed or gently rocked.
Disperse the cells by gentle pipetting (trituration) and then filter the cell suspension through fine mesh. Allow the cells to settle and decant excess liquid-containing enzymes; wash two to three times. Wash solutions containing FBS, BSA or other enzyme inhibitors may also be used to stop the enzyme digestion process. Re-suspend cells in appropriate medium or buffer and then quantitate cell yield and viability. It is important to quantitate the results of each dissociation step in order to effectively evaluate each procedure. The use of a cell counting chamber (hemocytometer) for yield quantitation and the use of trypan blue for viability quantitation are recommended. At this point cells can be seeded based upon recommended plating requirements or processed as desired.
Working with Enzymes
Lyophilized proteins are very hygroscopic, so they should not be opened in humid areas. Vials should be taken from the refrigerator at least 30–60 minutes before they are needed and kept in a desiccator. To be sure that enzymes are at room temperature make sure that the vial is not cool to the touch before opening. Most cell isolation enzymes can be repeatedly warmed to room temperature and returned, tightly closed, to a refrigerator, if the above precautions are heeded.
After being diluted with media or a buffer, proteolytic enzymes can undergo autolysis. Thus it is recommended that enzymes be dissolved immediately before use and stored on ice (2–8°C). They should not, however, be kept on ice for long periods; if necessary, they can be aliquoted and frozen at -20°C. Avoid too many freeze-thaw cycles for optimum stability. Special care must be taken with DNAses as they are prone to shear denaturation.
Generally, most of the enzymes used in cell isolation procedures (except trypsin) can be directly dissolved in a balanced salt solution or a buffer of choice (Figure 1). Stock trypsin solutions should be made by initially reconstituting the enzyme in 0.001 1N HCI to prevent autolysis prior to use. These solutions can be further diluted into the digestion medium or buffer immediately prior to use.
In many cell isolation procedures it is important to the survival of tissue during dissociation that the incubation medium be both well oxygenated and buffered at physiological pH. Both requirements are satisfied when the medium is equilibrated with 95% O2:5% CO2. Several balanced salt solutions contain the pH sensitive indicator dye phenol red. When red or purple in color, the medium is too alkaline. This sometimes occurs when the tissue is placed in the dissociation enzyme solution. Re-equilibration with O2:CO2 is usually necessary prior to incubation.
Gas should not be bubbled directly into any solution containing protein, as this can lead to frothing and denaturation of the protein with potential loss of biological activity. Gas can be sterilized by passage through a 0.22 micron membrane filter or through a sterile fiber plug such as those found in a sterile Pasteur or volumetric pipette. While mixing the solution containing phenol red, pass O2:CO2 continuously through the space above the liquid until the color indicates pH 7.2–7.4. Balanced salt solutions are often pre-gassed but should be equilibrated with sterile O2:CO2 each time the bottle is opened.
Buffered balanced salt solution will usually maintain constant pH regardless of the degree of oxygenation/carbonation and as a result can be easier to work with. Certain cell types are sensitive to particular buffer salts, which can have a negative impact on their viability. A review of published literature or references citing balanced salt solutions and media used for specific applications can be useful for selecting appropriate reagents.
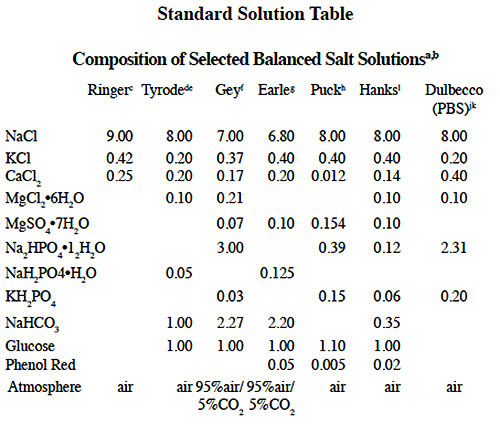
Figure 1
Releasing Cells
In order to transfer or pass cells in monolayer culture from one vessel to another it is necessary to release cells from the monolayer into suspension so that they can be easily handled by pipetting and diluting. Releasing cells from the monolayer is most often accomplished with purified trypsin in a procedure known as trypsinization. (Nontrypsin alternatives such as EDTA and animal-free proteases are also available for harvesting cells depending upon cell and cultureware type, extracellular matrices utilized, and application [Figure 2].)
Remove culture medium from cells and add sterile trypsin solution in a balanced salt solution or media; calcium-free Hanks (CFHBSS) is commonly used. Stock trypsin solutions should be made by initially reconstituting the enzyme in 0.001 1N HCI to prevent autolysis prior to use. These solutions can be further diluted into the digestion medium or buffer immediately prior to use. Allow trypsin to act on the monolayer for several minutes at room temperature or 37°C (or longer at 4°C). Remove trypsin solution gently to avoid disturbing cells and add balanced salt solution or media (often with serum or trypsin inhibitor to inactivate residual trypsin) and agitate vessel to disrupt monolayer and suspend cells. Carefully transfer cells.
Some researchers have found that procedures using purified trypsin or neutral protease can provide increased viability in cells after they are released compared to crude pancreatin or trypsin products. Viability is often determined by measuring cloning efficiency, which is defined as the ability of a single cell to attach to a culture vessel wall and divide to produce a colony of cells that is visible to the naked eye after staining. For this reason it is recommended that purified trypsin or neutral protease be used during trypsinization.
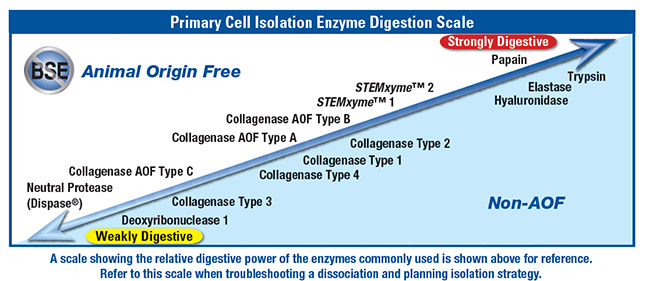
Figure 2
Summary
As lot-to-lot variation is typical in certain enzyme preparations, it may be important to pre-test a particular lot. A very practical approach to pretesting is to presample several different lots of enzyme at a time and select the best for your specific application. Working with a manufacturer or supplier who will support lot sampling will help ensure optimum results.
Carefully following the above procedures and guidelines will help provide consistent results and optimize yield, viability, and growth of desired cells.
Jim Zacka ([email protected]) is vice president of Worthington Biochemical, a manufacturer of animal origin free collagenases and other enzymes. For more information on cell culture, see the Worthington Tissue Dissociation Guide or http://www.labome.com/method/Cell-Culture-Media-A-Review.html.







