September 1, 2011 (Vol. 31, No. 15)
Guido Seidel, Ph.D.
Silvana Di Cesare, Ph.D.
Simple and Efficient Platform Approach Reduces Impurity Load for Downstream Processing
The use of E. coli for manufacturing therapeutic proteins is an economically attractive alternative to mammalian cell-based production. E. coli grows fast, has no demanding culture requirements, and produces high protein yields. Additionally, the purification process does not involve virus-clearance steps, which usually results in significantly lower costs per gram of product. These advantages make E. coli the system of choice for the production of proteins that do not need post-translational modifications.
Traditional E. coli expression involves the recovery of the product as intracellular inclusion bodies that require refolding. Frequently, the efficiency of the refolding step is very low and only small amounts of correctly folded protein are recovered.
Periplasmic expression in E. coli eliminates the need for refolding, as the proteins are correctly folded and/or assembled in the periplasm; however, fermentation yields are typically lower compared to inclusion-body processes due to the physical restriction of the periplasmic space.
To overcome these production limitations, Wacker Biotech has developed an E. coli expression technology termed Esetec® that secretes correctly folded proteins in high titers directly into the culture supernatant. This feature simplifies the bioprocess while rendering high yields of biologically active proteins.
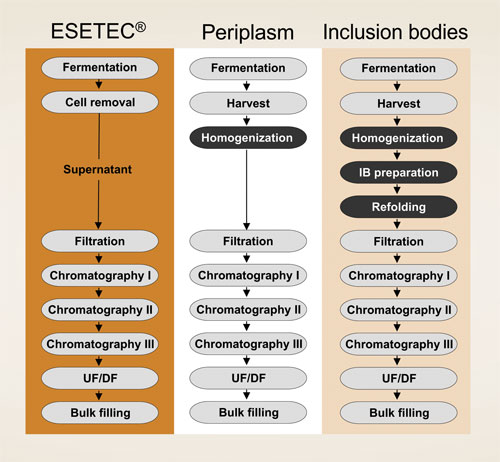
The high secretion efficiency of Wacker’s engineered strains, together with the lack of spatial constraint, results in superior yields of high-quality products at an affordable cost. Since soluble proteins are recovered in their native form from the culture supernatant, there is no need for cell disruption or refolding (Figure 1). This translates into simpler primary recovery and purification schemes, thus improving the overall process efficiency.
The primary recovery of recombinant proteins secreted into the culture supernatant usually encompasses centrifugation steps for cell separation and a series of filtration techniques for clarification, such as tangential flow filtration and depth filtration. The main goal of this filtration cascade is to remove cells or cell debris before starting the chromatographic sequence, to prevent clogging the first column, and to avoid unpredictable elution profiles.
Wacker has further developed and optimized the classical tangential flow filtration (TFF) to a simple and upscalable operation for primary recovery of proteins produced with Esetec strains. The supernatant containing the product is clarified by just a single tangential flow microfiltration step. This sole method efficiently removes all remaining host cells and cell debris, and also depletes large amounts of process-related impurities such as host-cell proteins, host-cell DNA, and endotoxins.
While these process-related impurities are efficiently removed, the product is recovered at high titers with negligible losses and the clarified supernatant is used without further conditioning for capture chromatography. By sparing additional conditioning steps such as dilution, depth filtration or diafiltration, the overall process performance is improved in terms of higher yields, minimized processing time, and reduced cost of goods.
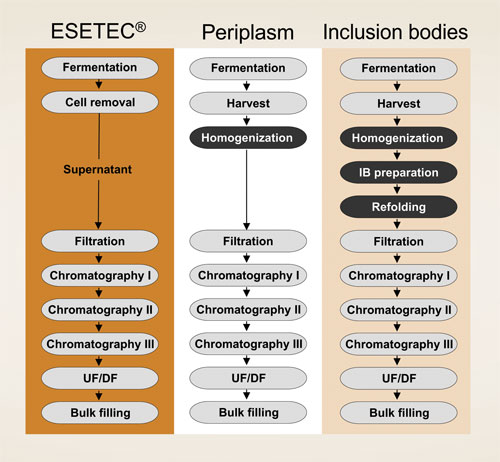
Figure 1. Protein secretion with Esetec reduces the number of process steps.
Primary Recovery
Wacker has established a simple and efficient platform approach for the primary recovery of proteins that are expressed and secreted by Esetec strains. The baseline fermentation is usually a fed-batch process in chemically defined mineral salt media, which is driven to a defined harvesting point by accurate µ-control and induction of expression with low IPTG concentration along with a shift to lower temperature.
On conclusion of fermentation, a balance is obtained between product quality, yield, and critical physicochemical and physiological parameters (e.g., nutrient concentration, viscosity, oxygen transfer, cell density, cell viability). The biomass is then removed by centrifugation at room temperature.
The load of impurities derived from Esetec cells in the supernatant is considerably lower than the amount of process-related impurities in whole-cell lysates from conventional E. coli processes. Therefore, it is conceivable that a single clarification step for Esetec processes might not only deplete the solids in the supernatant but also efficiently remove host cell-derived material. Removal of contaminants at this early stage would greatly simplify the subsequent chromatographic sequence, enabling higher product purity to be targeted while production costs are also conveniently reduced.
Tangential flow filtration has been implemented at Wacker as a sole clarification step. The process is performed with hollow-fiber modules. The modular nature of the TFF devices supports upscalability of the technology to accommodate larger volumes of supernatant. The extent of product recovery and impurity removal during TFF is based on the size and shape of the molecules. It, therefore, largely depends on the nominal pore size of the filter media and on the applied process parameters (e.g., shear rate and transmembrane pressure).
The pore size of the membrane (usually below 0.1 µm) and the most suitable operating parameters are identified for each product with a basic scouting approach early on during downstream process development (Figure 2). This is crucial if the TFF is to serve not only for clarification and volume reduction but also as a partial purification step.
Once cells have been removed, the culture supernatant can be used for filtration without further conditioning. The hollow-fiber units used for TFF are pre-assembled, ready-to-use, and disposable. Hence, material and equipment preparations are minimal, and there is no need to develop and validate cleaning procedures. These benefits help streamline the process by eliminating time-consuming and costly steps.
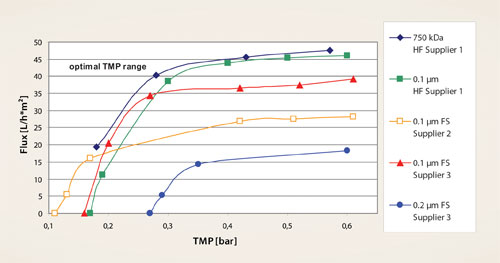
Figure 2. Scouting TFF experiments with hollow fiber (HF) modules and flat sheet (FS) cassettes: HF modules were tested at three shear rates (shown here: 15,000 s-1) and the FS cassettes at three feed flow rates (shown here: 1,200 L/h*m2). Hollow fibers showed higher flux rates than FS cassettes; the optimal operating TMP are highlighted.
The product yields in the TFF permeates are usually 85% (ranging from 70–90%), while host cell proteins are typically reduced by 35–45%. More than 90% of residual DNA and endotoxins are also depleted in this single step; clearance rates of 99% (2 logs) and 99.99% (4 logs) are commonly observed for residual DNA and endotoxin respectively (Figure 3, top panel). Altogether, the TFF permeates derived from Esetec cells consistently show improved impurity profiles compared to their counterparts from conventional E. coli (Figure 3, bottom panel).
Permeates from TFF can be used for chromatography (capture step) without further conditioning and are compatible with a broad variety of capture matrices. For example, clarified supernatants for Esetec-derived Fabs have shown superior performance in capture experiments using affinity chromatography, with product yields >90%. The permeates can also be loaded onto nonaffinity chromatographic media such as multimodal resins.
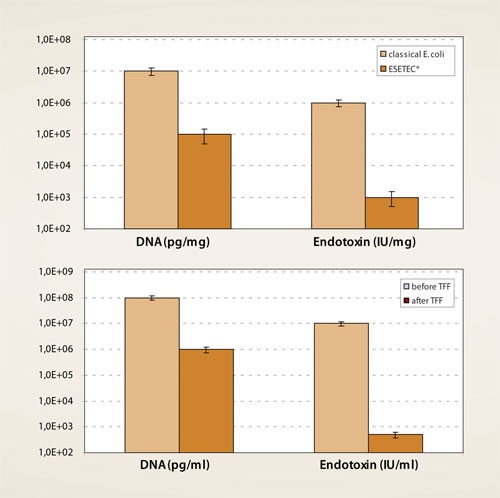
Figure 3. Top: Improved impurity profile of Esetec-derived cell culture harvests compared with lysates from classical E. coli strains (data after cell removal).
Bottom: A single TFF step efficiently depletes process-related impurities like host-cell DNA and endotoxin during primary downstream processing.
Process Costs and Product Quality
The use of an optimized TFF as a single clarification and partial purification step provides clear advantages over multistage operations for primary recovery. First, reducing the number of process steps lessens product losses that are unavoidable in multiple filtrations, thus increasing overall process efficiency while cutting operational costs. Second, the early depletion of impurities attained by TFF positively impacts the subsequent chromatography sequence in terms of performance and number of chromatography columns.
The use of fewer columns ought to significantly reduce costs; it is well known that the cost of goods for industrial bioprocesses is mainly driven by the number of chromatographic steps. Last, thorough depletion of impurities makes for higher purity of the final product, a key parameter that critically affects product safety.
For instance, the amount of endotoxins that E. coli brings into biopharmaceutical processes is considered the biggest drawback compared to non-E. coli expression technologies. Nearly all API specifications point to residual endotoxin concentration in the product as one key criterion when choosing the administration route.
Endotoxin specifications of Esetec-derived products are often below the LAL-test limit of detection (<0.5 EU/mg) and hence two to three times lower than classical specification set-points from conventional E. coli strains. Therefore, the combination of secretory E. coli expression and simple and efficient primary recovery might be a cost-attractive alternative for producing products otherwise produced in endotoxin-free systems.
The Esetec E. coli strain has been engineered to secrete heterologous proteins into the culture medium. This electron microscopy image shows a dividing Esetec® E. coli cell with budding secretory vesicles containing recombinant protein.
Guido Seidel, Ph.D., is head of process development, and Silvana Di Cesare, Ph.D. ([email protected]), is business development manager at Wacker Biotech, a wholly owned subsidiary of Wacker Chemie.