October 15, 2011 (Vol. 31, No. 18)
Junhai Kai
Aniruddha Puntambekar
David Sehy
Peter Brescia
Peter Banks Ph.D. Scientific Director BioTek Instruments
Microfluidics-Based Microplate Technology Offers Benefits Over Traditional ELISA Methods
Enzyme-linked immunosorbent assays (ELISA) are commonly used in research and clinical applications to quantify analytes from complex matrices such as serum and plasma/cell lysates. Excellent selectivity is provided by the use of a high affinity capture antibody and multiple wash steps. Good sensitivity is achieved through direct labeling of an antibody to a different epitope on the analyte or the use of a labeled secondary antibody. The basic ELISA workflow and sensitivity have not changed significantly in decades.
Siloam’s Optimiser microplate offers improved ELISA workflows, sample and reagent conservation, improved reaction kinetics, and the ability to fine-tune assay sensitivity through multiple analyte loading in fluorescent-based assays. The Optimiser uses an SBS-configured 96-well microplate architecture and a dedicated microfluidic channel immediately below each well (Figure 1) where binding occurs.
Each 200 x 200 μm spiral microchannel offers approximately 50 times larger surface area-to-volume ratio for capture of antibodies compared to a standard ELISA microplate, which relies on the microplate well bottom for capture. This allows for more efficient capture antibody binding and highly efficient antibody-antigen reactions, which decrease overall assay time.

Figure 1. Microfluidics microplate showing enlarged view of a microchannel associated with its microplate well
Principle of Operation
The Optimiser workflow (Figure 2) mirrors standard ELISA assay steps, however, the volumes used are significantly smaller, and washing steps are reduced.
Sample/reagent volumes as small as 5–10 μL are added to each well and drawn through the microchannel via capillary forces. Each microchannel, where binding occurs, holds 4.5 μL of liquid, and excess liquid is channeled to an absorbent pad under the microplate.
The sequence of reagent/sample additions is demonstrated in Figure 2. For each successive addition, the capillary barrier is broken at the microchannel inlet, and all previous reagents are flushed into the absorbent pad. Flushing excess liquid effectively removes unbound materials and also prevents cross-contamination of reagents within the microchannel. By making multiple additions of sample containing analyte, sensitivity can be tuned up to 100-fold higher than single additions of analyte.
Automating the process further enhances ELISA assay efficiency and increases throughput. An automated pipetting station, such as BioTek’s Precision™ Microplate Pipetting System, automatically loads analytes and reagents into the microfluidics microplate, and the multistation platform allows operation of multiple Optimiser microplates. Automation increases pipetting precision compared to manual methods, especially at the low volumes used in the Optimiser workflow, and also allows the user to attend to other tasks while the instrument is in operation.
As the Optimiser plate conforms to SBS recommended microplate specifications, it can be read in any standard fluorescence microplate reader.
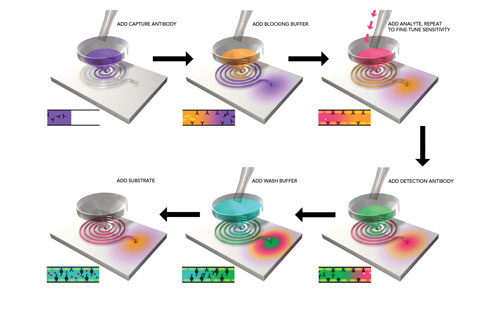
Figure 2. Key steps in the Optimiser workflow
Basic Comparison to Standard ELISA
When single aliquots of samples containing analyte are added to the Optimiser plate, detection and sensitivity performance, defined as a linear calibration curve slope, are similar to ELISAs. This analytical performance is suitable for many applications quantifying analyte in the pg/mL range. The benefit of using the Optimiser microplate for these applications is a significant reduction in reagent and sample consumption. Additionally, as wash steps are reduced, so is overall assay time, and sample throughput may be increased.
Increased Sensitivity
Some applications require higher sensitivity than standard ELISA detection limits. For example, cytokine secretions may be locally produced in high concentrations for autocrine and paracrine signaling, but in the supernatant of a microplate well, this concentration is greatly reduced and requires extremely sensitive detection.
For these applications, and others where the sample is dilute or at a very low concentration, the Optimiser technology and flow-through principle allow multiple repeat samples to be added per well to increase detection sensitivity.
To demonstrate this ability, interleukin-4 (IL-4) was used as an analyte and single concentrations spiked into cell culture media to simulate samples. These samples were loaded onto the Optimiser plate once at 10 μL, 20 times at 5 μL, and 100 times at 5 μL to construct calibration curves. The fluorescent-based assay was read on BioTek’s FLx800™ Fluorescence Microplate Reader with 528/20 nm excitation and 590/35 emission settings.
Left shifting of the calibration curves evident in Figure 3 implies increased analyte sensitivity. Each reagent addition, including sample, requires a five-minute incubation, so the overall assay time for a sample loaded once onto the plate is approximately 90 minutes. Samples loaded 20 times increase the overall assay time to approximately 3 hours, and samples loaded 100 times increase the overall assay time to approximately 10 hours.
The single addition of 10 μL of sample provides an operating range of 1–125 pg/mL, which is typical of a conventional ELISA-based assay. Yet this single load reduces the sample volume by 10-fold relative to a standard ELISA while retaining the ELISA’s operating range. Multiple small sample loads can significantly increase the sensitivity 10- to 100-fold and provide detection to fg/mL levels as shown in the Figure 3 table insert.
The novel microfluidics in the Optimiser microplate offers tangible benefits over traditional ELISA techniques for fluorescence- based assays. In addition to improved efficiencies and compatibility with existing assay kits and instrumentation, researchers can choose to use the plate to conserve sample and reagents, or to greatly enhance assay sensitivity. Both choices are further enhanced by automating the Optimiser-based assay to increase throughput, reduce operator error, and improve overall assay efficiency.
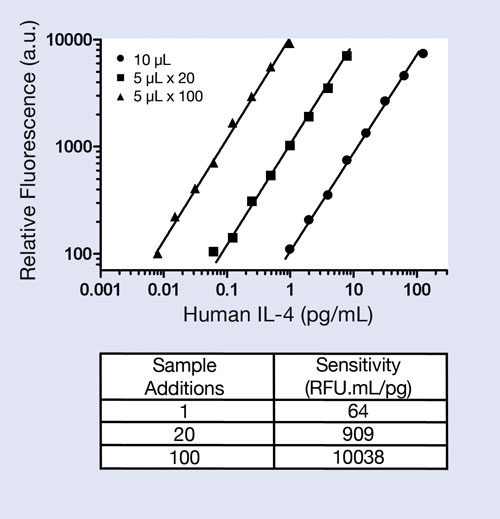
Figure 3. Human IL-4 assay in Optimiser ELISA microplate and sensitivity gains, defined as the slope of the calibration curves, is attributable to multiple sample loadings using the Optimiser plate.
Junhai Kai is director R&D, Aniruddha Puntambekar is vp, strategic planning and operations, and David Sehy is director, technology and business development (Western USA) at Siloam Biosciences. Peter Brescia ([email protected]) is applications scientist and Peter Banks is scientific director with BioTek Instruments.







