May 1, 2008 (Vol. 28, No. 9)
Barry Johnson
Johan Persson
Tom Thane Nielsen
Reshaping Reactions in Pharmaceuticals as well as Fine and Specialty Chemicals
Manufacturers of pharmaceuticals as well as fine and specialty chemicals face not only fierce competition but also growing safety concerns, tough environmental legislation, and rising energy costs. As a direct response to manufactures’ demands, Alfa Laval created a new technology, continuous plate reactors.
By integrating continuous reactions with advanced plate heat-exchanger technology, the plate reactor goes beyond batch reactor limitations, caused due to thermal inefficiency and other drawbacks, to enable safe, environmental, and cost-effective process improvements.

Table
How It Works
The plate reactor combines the properties of a continuous reactor with those of a plate heat exchanger. It contains flow-directing reactor plates that are sandwiched between heat-transfer plates, all held together by pressure plates to create a modular reactor unit (Figure 1).
The mechanical design of the reactor allows for use at elevated temperatures and pressures. Capacities range from 0.25 L/h up to 1 m3/h, covering all steps from laboratory-scale R&D to full production.
Multiple Advantages
There are several advantages of this technology, including the fact that combining the high heat transfer of a plate heat exchanger with efficient mixing produces reliable reaction control in one unit. This allows users to improve existing processes and develop new products.
In addition, unlike a batch reactor, the plate reactor handles a continuous flow of reactants. This makes it a safer proposition since the reaction volume is up to 99% smaller than that of a batch.
Also, unlike a batch reactor, which requires the dilution of reactants with solvents, a plate reactor can operate at an optimal concentration. This limits the creation of by-products and does away with solvent evaporation, which reduces energy consumption (Table).
The total benefit of the plate reactor depends, ultimately, on the process that is being run. For potentially dangerous processes, that a continuous plate reactor has a fraction of the hold-up volume of a batch reactor means that if something unexpected happened the effect would be limited. The possibility to monitor and control a continuous plate reactor ensures that any problem in the unit can be detected fast and stopped or controlled.
The layout of a plate reactor is not fixed; it can thus be configured with a different number of plates, have various measurement points, different inlets, and assorted flow paths for the utility and the process side. There is even the option to split it into two reactors in one frame depending on the process being run.
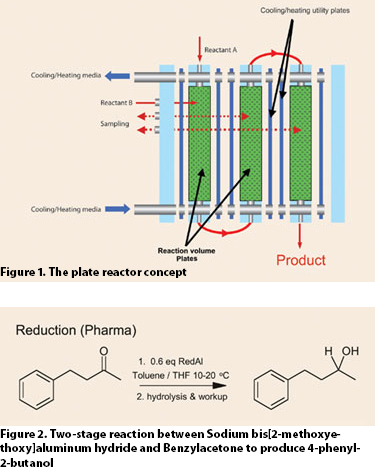
Figures 1 and 2
As a result of the heat-transfer capacity, highly exothermic reactions can be performed. This means that solvents can be reduced or reactions run at new conditions. With the higher operating temperatures and pressures, new possibilities for operating processes are facilitated.
Different zones can also be established along the reaction channel. This means that a chemical manufacturer can perform different reaction steps in a single unit, reducing both equipment needs and process set-up.
Cleaning and Inspection
In any hygienic process, the ability to clean and inspect is essential. The plate reactor can be disassembled and reassembled easily, facilitating thorough cleaning and reducing downtime. The flow through the unit is almost ideal plug flow and combined with good mixing, can reduce unwanted side reactions.
Batch Reactor
When using a batch reactor to perform a reduction reaction (Figure 2), a typical operating procedure includes many steps. These steps include filling the reactor with reactant and solvent and then cooling to 0°C, slowly adding excess of the reduction agent, RedAl, over 2–4 hrs while cooling to keep the temperature and the reaction rate low, and then more mixing and cooling. For the hydrolysis step, this procedure is largely repeated. Then, users filter off solid waste, perform product separation, and end up with a 80–90% yield of the purified product.
In a standard batch operation in a 1 m3 stirred-tank reactor, the reactions will take place over hours compared to the plate reactor, where the reaction takes place in seconds.
Plate Reactor
A plate reactor with three plates was used for comparison with a batch reactor. All of the plates are connected in series and operated at the same temperature; water was supplied as the utility fluid from a simple mixer tap.
RedAl solution was passed through the first plate to establish the reaction temperature. At the first port of the second plate, benzyl acetone was added, and the reaction was initiated. In the final plate, the hydrolysis solution was added. Pressure was adjusted to prevent too much vapor or boiling.
Using thermocouples placed in the remaining reactor ports (Figure 3), the progress of the reaction was viewed. In the second reactor plate, where the RedAl reaction took place, there was a temperature peak corresponding to the reaction heat. The peak was sharp and close to the feed point—indicating an almost instantaneous reaction.
Despite this, the yield was almost quantitative. During operation, the system was also able to handle the hydrogen gas evolved from the hydrolysis of excess RedAl.
Figure 3 shows that isothermal operation is not necessary; problems associated with side reactions or thermal degradation are often caused by the combination of high temperature and a long process time. As there is no holding of intermediates here—they are simply made and then used in successive parts of the reactor—the reaction is allowed to go as fast as it wants, resulting in only a few seconds residence time in the plate reactor.

Figures 3 and 4
We determined the approximate kinetics for the two reaction steps from a batch pre-study and these were validated using the results above. This information was then used to simulate the scale-up options for the process within a larger plate reactor.
The plate reactor (Figure 4) has shown many significant advantages including a >99% conversion, no by-products seen by GC/MS, lab-scale operation yielding 5 mol/hr (0.7 kg/hr) of alcohol product, and achievement of maximum yield in the plate reactor in less than 10 hours.
By expanding production engineers’ knowledge about reactions, this new technology enables pharmaceutical and fine and specialty chemical manufacturers to transition from batch processing to continuous production.
—
Barry Johnson, Ph.D., is process development manager, Johan Persson is PR specialist, products and applications ([email protected]), and Tom Thane Nielsen is market communication manager, food technology and life science, at Alfa Laval. Web: www.stepintoart.com.