January 1, 2012 (Vol. 32, No. 1)
Hemanth K. Potluri
Satish C. Tripathi, Ph.D.
The Biologics Price Competition and Innovation (BPCI) Act, a subtitle of the Affordable Care Act (ACA), amends the existing policy on biological drugs, the Public Health Service Act (PHS), to create a provision for the rapid approval of biosimilar drugs.
The FDA defines “biosimilar” as a drug that is “highly similar to the reference product, notwithstanding minor differences in clinically inactive components,” with “no clinically meaningful differences between the biological product and the reference product in terms of safety, purity, and potency.”
In this definition, purity refers to the absence of extraneous material that may or may not have an effect on the efficacy of the drug, and potency refers to how powerful the effect of the drug is on the body. While this may seem very much like the definition for a generic drug, there are a few fundamental differences between the two types of copycat drugs, which will be explored in this article.
A company creating an innovator drug has already invested heavily in R&D and compiled sufficient data to prove the effectiveness of the active ingredient. Therefore, the FDA created an expedited pathway for approving generic drugs under the Hatch-Waxman Act. The law states that for a generic to be approved, it must be shown that it is pharmaceutically equivalent (same active ingredients, route of administration, and strength), as well as bioequivalent, meaning that there is a statistically insignificant difference between the amounts of each drug that become available to the active site when the same dosage is used.
Lacking the cost of drug discovery, generics manufacturers are able to dramatically decrease prices, in some cases by up to 80%.
The reason the FDA can stipulate that generics must be the same as their reference products is that generics are small molecule drugs. Members of this class of drugs are chemically synthesized and are relatively simple and stable. Biologic drugs, in contrast, are comparatively large molecules made from living sources. Early biologics were harvested from animal sera, while newer ones are manufactured by recombinant DNA technology.
The manufacturing processes are vastly more complex than those for small molecule drugs and introduce many more variables, such as the species and strain of host cells, and the quality of the purification process. This makes it impossible to prove that two biologics are the same.
Unsurprisingly, it requires more extensive testing to show that the reference product and the biosimilar will have the same clinical effect than for generics. The BPCI Act mandates studies in vitro and in vivo, as well as clinical studies to demonstrate that the biosimilar is alike enough to the innovator and is safe for patients. Interestingly, it also gives the FDA the power to deem one of the above tests unnecessary.
The FDA will implement this law and draft guidelines, allowing biosimilars to begin to be approved in the U.S as they already are in Europe and Japan. There is much debate over what the outcome of this decision will be.
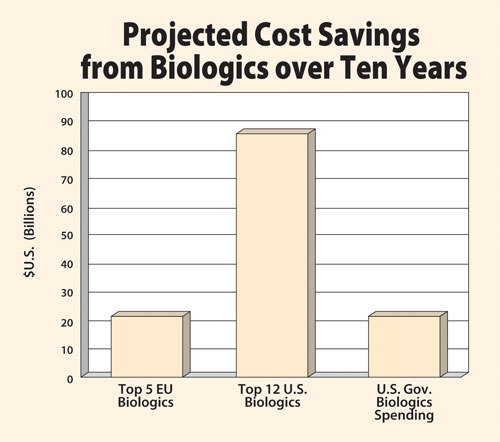
If biosimilars were available to replace biologics, the savings would be considerable but vary regionally. The savings shown for the top five EU biologics were projected by the European Union. Economist Robert Shapiro provided data for the top 12 U.S. biologics. U.S. Government biologics spending was obtained from the Congressional Budget Office.
Opportunities
The additional required studies along with higher overall manufacturing costs of e50 to e80 million (about $71–$113 million) per drug of biologics cause the European Generics Medicines Association (EGMA) to predict only 20–30% cost savings. However, the EGMA also noted that a 20% price reduction in five biologic drugs would save the EU over e1.6 billion ($2.3 billion) a year.
Economist Robert Shapiro estimates that biosimilar versions of the top 12 categories of biologics with expiring patents will save the U.S. $67–$108 billion over 10 years. The Congressional Budget Office expects that the BPCI Act will cause the following over a ten-year period: a $25 billion savings in general expenditures on biologics; a $5.3 billion increase in federal spending to implement the law; but, a simultaneous decrease of $5.9 billion in direct federal spending on biologics.
This article will weigh the benefits and potential pitfalls of developing this new type of drug. With the correct modifications to the existing drug access system including government programs, and private sector initiative, biosimilars could greatly increase access to biological drugs.
Challenges
Although the BPCI Act has a provision for declaring a biosimilar interchangeable with its reference, it is unlikely that many drugs will achieve this designation. In the EU, automatic substitution is banned because of the dangers of switching between similar biologics, for example, switching between brands of antiepileptic drugs can cause seizures.
The largest fear that many have is that biosimilars will induce immunogenic reactions. The formulation of the erythropoietin biologic Eprex (indicated for anemia caused by chronic kidney disease and chemotherapy) was slightly modified, causing the formation of neutralizing antibodies that reacted with endogenous erythropoietin. This caused red blood cell aplasia, meaning that the bone marrow ceased to produce red blood cells.
Drugs such as Eprex that can suddenly become dangerous due to minute differences between batches raise dire safety concerns for follow-on biologics, which will have unavoidable differences from the original. Aside from the more dramatic side effects, immunogenicity can also cause reduced efficacy and delayed serum sickness-like symptoms.
One study found that by using a battery of assays, the biosimilars Binocrit and Retacrit had comparable quality to Eprex and Dynepo. However, they also found significant differences in potency from what was printed on the packaging, which could create problems in prescribing the correct dosage for patients.
Another study focused only on epoetin alpha raised concerns about the higher level of mannose in the biosimilar and the need for more extensive and sensitive studies to detect any possible clinical implications.
Even in the small molecule field, generic forms of brand-name drugs can have potentially clinically meaningful differences. According to one study, generic paclitaxel and brand name Taxol have significant potency differences. Though both inhibit the growth of cancer cells by arresting their development in a specific stage of mitosis, the generic had a higher severity of these effects.
This is problematic because the FDA mandates that all generics be clinically equivalent to their references at the same dosage. When this is not true, the interchangeability of the two drugs is called into question.
The R&D cost of a new drug is estimated to be about $1.2 billion. Biopharmaceuticals manufacturers claim that building manufacturing facilities costs up to $450 million more. Without adequate mechanisms to recoup this cost, the incentive to bring new innovative drugs to the market is greatly reduced.
For this reason, the BPCI Act grants a 12-year exclusivity period during which the innovator company does not have to share its clinical data and no biosimilars can be approved. Interestingly, this development cost figure varies widely from source to source.
Shapiro claims that the average cost is only $530 million. Groups like the AARP, the American Medical Student Association, and the National Women’s Health Network, support a five year exclusivity period. This would allow more competition and bring down the prices of biologics much more rapidly.
In addition, it is the same period of exclusivity permitted for small molecule drugs before generics can be made. Biosimilars and generics manufacturers argue in favor of a reduced exclusivity period, claiming that the competition will encourage innovator companies to create new biologics in order to continue to profit.
One biosimilar company Hospira, also states that in Germany, where legislation is more favorable to biosimilar companies, the company has seen a 20–30% price drop in erythropoietin drugs because of competing biosimilars. For this reason, President Obama proposed a compromise of seven-year market exclusivity in his 2012 budget proposal. This proposal will be seriously debated by advocates of both sides as it could potentially compromise return on investment by innovator companies.
Hemanth K. Potluri ([email protected]) is a consultant and Satish C. Tripathi, Ph.D., was president at Biomedical Consulting International.







