June 1, 2010 (Vol. 30, No. 11)
Kathy Liszewski
Nanoparticle Carriers and Noninvasive Methods Are Among Recent Advances
Despite considerable challenges, RNA interference (RNAi) as a therapeutic strategy continues to draw excitement and scrutiny. Two recent meetings—Select Biosciences’ “RNAi and miRNA World Congress” and “RNAi 2010”—provided updates on the field. A number of presentations focused on advances in the use of major players in silencing the double-stranded RNA segments termed small (short) interfering RNAs (siRNA). Research highlights included novel ways to stabilize and deliver siRNA, especially the use of nanoparticles for targeted delivery.
“Although the initial buzz about RNAi came from its successes in cell cultures, achieving acceptable efficacy in vivo was another matter,” according to Dmitry Samarsky, vp, technology and development, RXi Pharmaceuticals.
“There are many obstacles facing successful RNAi compound delivery,” Dr. Samarsky explained. “If introduced systemically—that is, into the circulation—compounds are often very labile in biological fluids. They can be quickly inactivated by blood nucleases or cleared from the body. Even when directly injected into tissues of interest, there is no guarantee of delivery inside the target cells.
“It is hard, for example, for an RNAi molecule to cross the cellular membrane, and there are interstitial and cellular nucleases as an added challenge to deal with as well. If successful at penetrating the target cell, the next question is whether they will be able to activate the RNAi induced silencing complex. In addition, RNAi molecules can evoke an undesired immune response through multiple mechanisms.”
To address these multifaceted problems, RXi developed a class of RNA molecules called self-delivering RNAi compounds, or sd-rxRNA™. “Our scientists focused on a strategy that combined particular oligo configurations and chemical modification patterns. These specially designed molecules help resolve many problems associated with therapeutic RNAi delivery,” Dr. Samarsky added.
“They have enhanced nuclease resistance, remain active longer in the circulation, facilitate organ and tissue delivery, and stimulate spontaneous cellular uptake. Because we have chemically modified and optimized the content, they avoid immune responses and increase RNAi specificity.”
Part of the design strategy was to reduce the size of the molecules. “Most conventional siRNA duplexes are between 19 and 25 base pairs. Ours contain duplexes of less than 15 base pairs. We found that reducing oligo content greatly enhances their ability to cross the cell membrane.
“The sd-rxRNAs seem to operate via a universal mechanism and are taken up by various cell types in vitro. It is very fast. Fluorescent molecules can be observed within minutes of administration. The next step for RXi was to assess and verify activity in multiple cell types in vivo. We have already demonstrated the uptake of the molecules by cells in skin, lung, and eye after local administrations, as well as by the liver upon systemic injections. We are continuing to examine applications.”
Image-Guided siRNA Delivery
As RNAi gains momentum as a therapeutic strategy, new noninvasive methods for detection of siRNA delivery and verification of silencing become paramount. Zdravka Medarova, Ph.D., assistant professor, Massachusetts General Hospital and Harvard Medical School, and colleagues have developed dual-purpose nanoparticle probes that not only facilitate in vivo transfer of siRNAs but also simultaneous imaging of their accumulation in tumors via magnetic resonance imaging (MRI) and near-infrared optical imaging (NIRF).
“Our probes have four major advantages,” Dr. Medarova reported. “They can be detected noninvasively, increase circulation half-life, enhance versatility, and hone in on their target. They consist of magnetic nanoparticles labeled with a near-infrared dye and conjugated to siRNA molecules specific to therapeutic targets. These nanoparticles have excellent biodistribution and blood half-lives. The half-life in humans is about 24 hours and in mice 10–12 hours. Additionally, we have seen no signs of any systemic toxicity.”
Aside from engineering for easy detection via MRI or NIRF, special targeting features can also be included. “We can modify the nanoparticles to include antigen targeting or membrane translocation modules for cell-specific intracellular delivery. Imaging allows in vivo tracking of the tumor uptake of these probes. We demonstrated this in two separate tumor models.”
Dr. Medarova said since her initial findings there has been an explosion of additional studies. “siRNA delivery via nanoparticles is becoming a trend. Many companies have shown an interest in this type of technology.”
The next phase of her studies is to further develop coated nanoparticles for miRNA applications. “The idea is to stop the metastatic processes at very early stages, at the level of epigenetics. We hope to exert such control via our technology.”
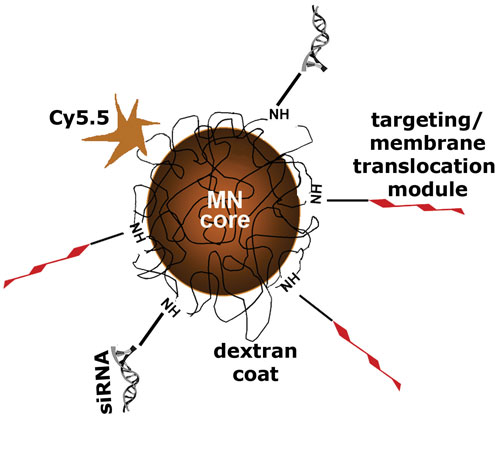
Scientists from Massachusetts General Hospital and Harvard Medical School have developed nanoparticle probes that facilitate in vivo transfer of siRNAs. The probes consist of magnetic nanoparticles (MN) labeled with a near-infrared dye (Cy5.5) and conjugated to antigen targeting or membrane translocation modules, as well as siRNA molecules specific to therapeutic targets.
Attacking Cancer
Effective siRNA therapy against cancers must not only target the primary tumor but also must reach metastatic lesions, indicated Esther Chang, Ph.D., professor, Georgetown University Medical Center. She also employs nanotechnology and has developed a tumor-targeting approach to attack both.
“Solid tumors are difficult to treat because you must deliver the therapeutic systemically and still penetrate deeply enough into the tumor through very small blood vessels to treat the entire tumor,” she explained. “Moreover, if the treatment doesn’t also reach distant metastatic tumor cells, there will be continued cancer growth.
“We developed a platform nanodelivery system composed of self-assembled, biodegradable, cationic liposomal nanoparticles with targeting molecules that home to receptors prevalent on cancer cells such as the transferrin receptor. The nanocomplex systemically delivers siRNAs and other nucleic acid-based molecules to the target. The nanoparticles also can be engineered to carry diagnostic MRI contrast agents and small molecules.
“It was quite exciting to find that this strategy could reach both primary and metastatic tumors. We also verified that this nanotechnology platform fulfilled the three key requirements of effective tumor-targeting delivery: demonstrated presence of the therapeutic payload in the tumor, localization only to tumor and not to normal tissue, and a dose-dependent presence of the therapeutic payload in the tumor cells. We have performed studies using this approach to deliver various therapeutic payloads including siRNA in 15 to 16 solid-tumor models such as melanomas, breast, prostate, pancreatic, cervical, and lung cancers.”
Significantly, she also found that these nanocomplexes can dramatically sensitize human tumors in these mouse models to radiotherapy and chemotherapy. Long-term tumor elimination and prolonged life span of the animals was observed.
Additionally, Dr. Chang has utilized her platform for delivery of the human tumor suppressor gene, p53. Those studies are completing a Phase I trial as a single agent. “So far minimal side effects have been noticed and in some cases, there have been significant tumor responses, even at the lowest dose.
“This is truly a platform technology. For example, incorporating the imaging agents into this 100 nanometer complex allows much greater sensitivity of detection and visualization of even minute metastatic lesions in the lungs,” Dr. Chang said.
What is the next challenge? “Funding! We hope to enlist the help of pharma to carry out further clinical studies.”
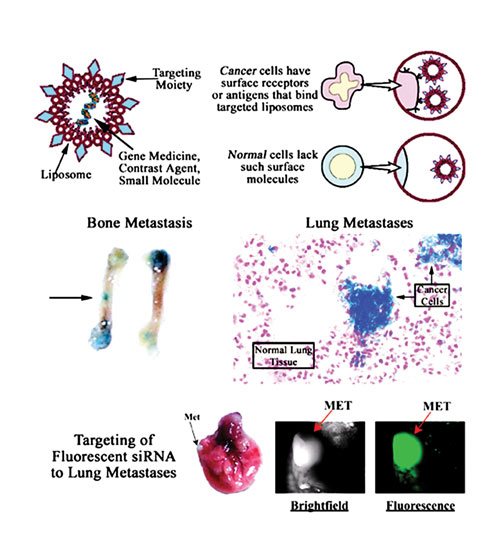
Selective targeting of tumors by systemic delivery of nanocapsules [Georgetown University]
Cocktail Therapeutics
Human diseases often represent complex pathological processes associated with abnormal gene expression. Although single-drug treatments may provide significant therapeutic benefits, disease progression or development of drug resistance can circumvent the effect of a single drug. Sirnaomics is pursuing a strategy to overcome such hurdles utilizing a combination of multiple drugs to gain a stronger therapeutic efficacy.
“siRNAs are ideal components of therapeutic cocktails because of their chemical homogeneity. They have the same four nucleotides, the same charge/hydrophobicity for each sequence irrespective of the target gene, the same source of origin, and a common, reproducible manufacturing process,” Patrick Lu, Ph.D., president and CEO, said.
“Our approach is to use multitargeted siRNA cocktails with nanoparticle-mediated delivery to enhance therapeutic efficacy. But, the multitargeted siRNA cocktail needs to be logically designed and able to work in vivo. We use a proprietary algorithm to develop a Tri-blocker design for siRNA duplexes that targets up to three disease-causing genes in one cocktail.”
For delivering the cocktail, the company continues to develop three generations of nanoparticle systems. “The first generation nanoparticle is based on a self-assembling property in which cationic liposomal or polymeric materials are mixed with siRNA molecules,” Dr. Lu said. “The second-generation nanoparticle is chemically modified with specific ligands to target desired tissues or cells. The third generation of nanoparticles, developed in house, is a silica-based nanoparticle system comprising an up-conversion core coated by a photosensitizer drug and siRNA. It can be temporally and spatially controlled with near infrared activation.”
According to Dr. Lu, third-generation nanoparticles are especially valuable for the treatment of cancers. “An infrared light is used to excite up-conversion nanoparticles to produce visible light, which activates the photosensitizer on the nanoparticles to produce singlet oxygen. This then destroys and weakens the cell membrane, promoting the delivery and release of siRNA molecules into the cytoplasm. This strategy provides an effective treatment of cancers, especially those located deep within the human body.”
Dr. Lu noted that the technology can be used in cancer applications including hepatocellular carcinoma, renal carcinoma, non-small-cell lung carcinoma, breast carcinoma, and pancreatic carcinomas. Preclinical studies are ongoing for each of the nanoparticle systems.
Piggyback Therapeutics
Like the fabled Trojan horse, siRNA entry into the cell’s interior requires some creative approaches. Paul White, Ph.D., senior lecturer at the Monash University Institute of Pharmaceutical Sciences in Melbourne, Australia, and his colleagues are utilizing one of the most abundant carrier proteins in the blood—albumin—to ferry siRNAs attached to nanoparticles across the vascular endothelium.
“Our overall project focuses on the cardiovascular system,” he reported. “We have some targets that are involved in damaging reactive oxygen species production in cardiovascular disease and will be testing whether siRNA-albumin delivery can reduce the damage done to the heart and blood vessels in a model of high blood pressure. However, there are a number of other organs for which a delivery system capable of allowing siRNA extravasation would be valuable.”
Dr. White described the technology as a piggyback approach that capitalizes on the property of albumin to penetrate cardiac and vascular interstitium. “We have utilized both covalent conjugation and nanoparticle construction approaches. For the covalent conjugation we employed a heterobifunctional linker to join amine-modified siRNA with albumin, via an exposed cysteine residue. We used fluorescently labeled siRNA and showed extravasation of these agents. We have also shown knockdown in mouse hearts using real-time RT-PCR. For the particle strategy, we used a cationic lipopeptide containing lysine residues to provide positive charge and histidine residues to aid endosomal escape.”
Dr. White’s future studies will continue to refine and better understand the system. “We are on the way to a robust generic approach to delivery of siRNA across the vascular endothelium; we need to understand better the scope of the albumin-mediated delivery mechanism.
“The issue of endosomal escape is another that we are working on at the moment. The hard part of siRNA delivery is not only addressing each individual barrier, but doing so in a way that does not bring other problems into play. Success here requires a thorough understanding of siRNA fate in plasma, route to the target cell, and activity inside the cell,” Dr. White concluded. “Gaining this understanding is a slow process, but we are getting there.”
Naked siRNA Delivery
Delivery of siRNA is not a major hurdle for every organ and tissue, noted Elena Feinstein, M.D., Ph.D., CSO at Quark Pharmaceuticals. “We decided to examine which cells and tissues were amenable to efficient delivery of naked (i.e., substantially nonformulated) siRNA and then looked for diseases involving these cells.”
Just as important to choosing the optimal target and indication, the company also worked to develop a strategy for designing and then chemically stabilizing the siRNA. “We were able to stabilize and improve the drug properties of siRNA therapeutics by using specific chemical modifications.”
Dr. Feinstein cited the example of siRNA and certain kidney diseases. “After intravenous administration, Quark synthetic siRNAs are rapidly filtered by the kidney and then some of the siRNA is reabsorbed by the proximal tubules.”
Proximal tubules represent the kidney substructure that is specifically affected in the majority of diseases associated with acute kidney injury.
“This prompted us to focus on indications involving acute kidney injury and on systemic administration of naked siRNA as a delivery mode. We found that we can administer naked siRNA intravenously and readily see a therapeutic effect in rat models of ischemia-reperfusion injury in kidneys.”
The company’s QPI-1002 is the first systemic siRNA drug to enter clinical trials. It targets p53 mRNA and currently is in development for prevention of acute kidney injury after major cardiovascular surgery and for prophylaxis of delayed graft function in kidney transplant patients. Phase I/II trials are fully enrolled and are currently being completed. The plan is to start Phase II studies later this year.
“There are other target tissues that are also suitable for naked siRNA therapies such as the eye, where many cell types can efficiently take in chemically modified siRNA, but in particular retinal ganglion cells,” according to Dr. Feinstein. “We have developed an siRNA that can be directly injected into the eye as an ocular neuroprotective agent. The initial clinical trials will focus on treating an acute ischemic disorder of the optic nerve called nonarteritic anterior ischemic optic neuropathy. Those studies are in Phase I trials. Our partner, Pfizer, is currently conducting two Phase II trials with PF-4523655, another siRNA developed by Quark, for diabetic macular edema and wet age-related macular degeneration.”
Overall, optimism remains high that the barriers to the successful delivery of siRNA will be overcome. The most positive signs for the future of siRNA-mediated gene silencing are emerging studies showing knockdown with low doses of siRNA in complexes or conjugates that target specific cell types.

Localization of siRNA in rats following delivery by inhalation, intravenous, or intravitreal injections, respectively. Left: fluorescent microscopy (FM) visualization of siRNA in tissue specimen. Right: light microscopy images of ISH detection of siRNA in tissue. Oligo probes for siRNA detection in the lungs and kidneys were DIG-labeled, whereas probes in the retina were labeled with 33P (black dots). A: alveoli, B: bronchioli, PT: proximal tubules, GCL: ganglion cell layer, INL: inner nuclear layer, ONL: outer nuclear layer. [Quark Pharmaceuticals]



