February 1, 2009 (Vol. 29, No. 3)
Steven M. Riddle
TR-FRET Tools Broaden the Potential of Drug Screening
Over the past decade, small molecule kinase inhibitors have emerged as promising therapeutics, particularly in oncology. Through data that has emerged during this period, it has become increasingly apparent that small molecule inhibitors interact differently with the various conformational and activation states of a protein kinase.
For example, the FDA-approved kinase inhibitors Gleevec®, Nexavar®, and Tykerb® preferentially interact with nonactive kinase conformations, and such modes of binding can lead to increased specificity due to compound interactions with less-conserved residues that are accessible only in nonactive forms of the kinase. In other cases, the preferential binding of small molecule inhibitors to nonactive forms of a kinase can be accompanied by slow compound “off rates”. Slower off rates can lead to prolonged biological effects, which in turn can boost the overall efficacy of the inhibitor (an observation reported for Tykerb).
Challenge of Traditional Assays
An understanding of the diverse binding modes of kinase inhibitors and their effects on biological systems can provide researchers with important clues regarding the effective design of new drugs.
At this time, however, efficient, broadly applicable high-throughput screening (HTS) analysis of kinase inhibitors is primarily achieved through kinase activity assays. Activity assays, by their very nature, are only useful for measuring inhibition of active kinase preparations, which means that analysis is limited to active targets for which suitable in vitro substrates have been identified.
An alternative to the activity assay involves directly measuring the binding of small molecules to the kinase under investigation. Such binding assays can be used to study kinases prepared both in active and nonactive states. This allows the researcher the freedom to choose the specific kinase state to be targeted in HTS campaigns. Moreover, the mechanisms of newly identified inhibitors can be evaluated by measuring binding characteristics to multiple states of the kinase target.
Simple TR-FRET Binding Assay
Invitrogen’s LanthaScreen™ Eu Kinase Binding Assay platform was designed for HTS, lead optimization, and mechanism of inhibition (MOI) studies using kinases in either an active or nonactive conformational state, and to be broadly applicable across the kinome. The assay measures the binding and displacement of an Alexa Fluor® 647 dye–labeled kinase inhibitor (tracer) to a kinase active site.
Bound tracer is detected by addition of a europium (Eu)-labeled antitag antibody that specifically labels the kinase of interest. When the tracer is bound, fluorescence resonance energy transfer (FRET) occurs between the Eu-labeled antibody and the tracer; when the tracer is displaced by the bound inhibitor, FRET is dramatically reduced (Figure 1).
In addition to the benefits of flexible target choice, this assay format offers a robust time-resolved–FRET (TR-FRET) readout, simple assay optimization and setup, and the ability to use low concentrations of kinase (in the 0.1 to 5 nM range). Because the tracers are ATP-competitive, the assay will detect any compound that binds to the ATP site. These compounds include Type II inhibitors such as Gleevec, Nexavar, and BIRB-796, which bind to the ATP site, as well as an adjacent allosteric site, exposed in nonactive states of some kinases.
Using a small set of broad-spectrum tracers, a Eu–anti-GST antibody, and a new high affinity Eu–anti-His antibody, assays have been developed for more than 211 of the 300 kinases in Invitrogen’s portfolio (Figure 2). This broad coverage includes more than 30 kinases for which robust HTS activity assays have been difficult to develop. These newly tractable targets include DDR1, which was recently identified to be a cellular target of Gleevec, as well as CDK8, and TGFBR1 (ALK5) (Figure 3).
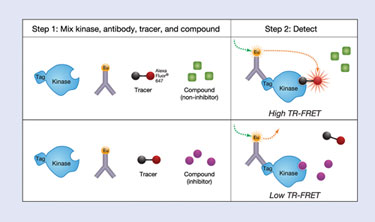
Figure 1. LanthaScreen Eu Kinase Binding Assay
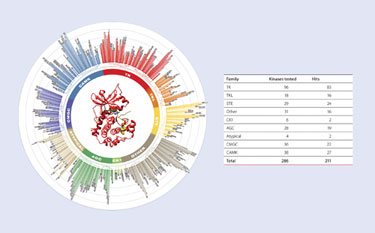
Figure 2. Kinase coverage
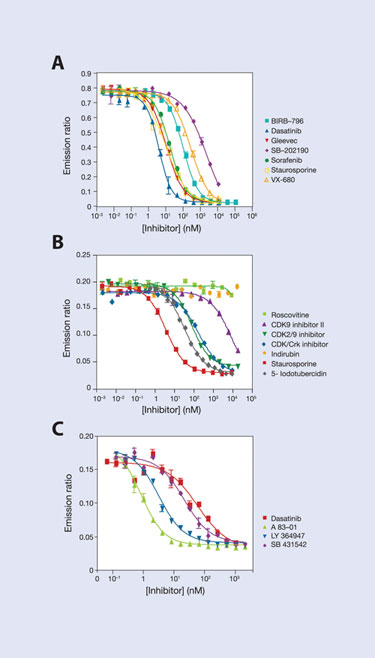
Figure 3. IC50 curves for several kinase inhibitors for (A) DDR1, (B) CDK8, and (C) TGFBR1 (ALK5)
Multiple Classes of Kinase Inhibitors
The first FDA-approved kinase inhibitor, Gleevec, was discovered in cell-based assays and later shown (through biochemical assays) to bind preferentially to a non-phosphorylated (nonactivated) form of Abl. The LanthaScreen Eu Kinase Binding Assay has been used to demonstrate tight binding of Gleevec to dephosphorylated Abl and weaker binding to active (phosphorylated) Abl (Figure 4); Gleevec-bound phosphatase-treated Abl has nearly 40-fold higher affinity than untreated Abl (the affinity of the control compound staurosporine was similar for the two forms).
These experiments demonstrate the flexibility of this binding assay for analyzing kinase inhibitors—it is possible to modify the phosphorylation state of a kinase by adding a phosphatase, and, in many cases, another kinase, without the need to repurify the target kinase prior to analysis. This is in contrast to activity assays, for which contaminating phosphatase and kinase activities would be much more likely to interfere with analysis.
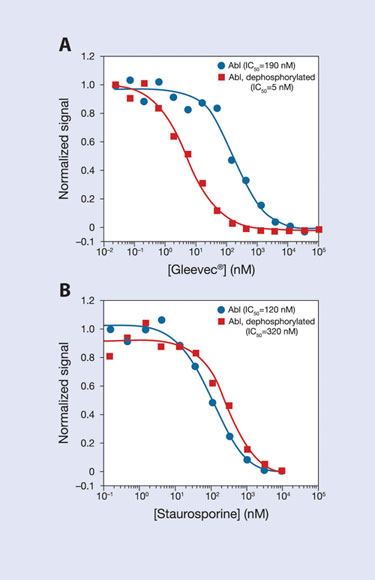
Figure 4. Preferential binding of the Type II inhibitor Gleevec® to dephosphorylated Abl.
Assay the Right Kinase
A challenge often faced when developing kinase activity assays, especially for less-studied targets and those with low specific activity, is ensuring that the measured activity is due to the target of interest and not to low levels of contaminating kinases from host cells.
This is particularly true when using generic substrates and when specific control inhibitors are unavailable. This complication is removed in the LanthaScreen Eu Kinase Binding Assay, as the signal is derived only from an epitope-tagged kinase and is not affected by low levels of any contaminating kinase or phosphatase activities.
Kinetics of Compound Binding
In addition to enabling screening of different enzyme forms, the LanthaScreen Eu Kinase Binding Assay also has the advantage of allowing real-time measurements of compound binding, unlike activity assays. This feature can facilitate characterization of many so-called allosteric or Type II kinase inhibitors that often have a slow “on rate”, which arises when the compound binds preferentially to a conformation of the kinase that is in equilibrium with other forms. Although this can be measured by monitoring the activity of the kinase at various time points after preincubating the kinase and inhibitor, it is simpler to monitor this binding directly, as shown in Figure 5, in which the binding of BIRB-796 to p38 alpha is monitored over time.
The LanthaScreen Eu Kinase Binding Assay platform enables researchers to assay previously challenging targets, whether for inclusion in broad selectivity studies or for primary and secondary screening. Unlike traditional activity assays, this novel format allows for rapid, efficient assessment of inhibitor binding to kinases in either active or nonactive conformations, as well as kinases with activity too low to be easily studied with traditional methods.
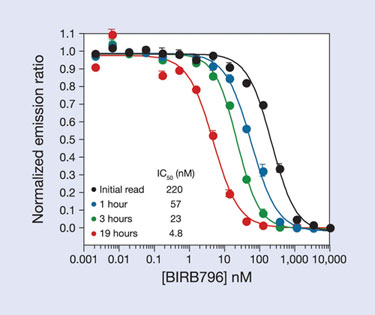
Figure 5. Slow binding of the Type-II kinase inhibitor BIRB-796 to p38 alpha
Steven M. Riddle (steve.riddle@invitrogen. com) is a senior scientist at Invitrogen. Web: www.invitrogen.com.



