January 1, 2009 (Vol. 29, No. 1)
Ilene Schneider
Firm Seeks to Improve Diagnosis and Treatment of Tuberculosis and Bacterial Pneumonia
Today, about one-third of the world’s population, or two billion people, carry the tuberculosis (TB) bacteria, although most never develop active TB disease. Studies estimate that 65 to 70 million people have active TB, there are 8 to 10 million new cases of active TB per year, and there are 2 million annual deaths.
Carol Nacy, Ph.D., became aware of the unmet medical needs of TB while working at the NIH. She knew that, in 1993, WHO declared tuberculosis to be a global health emergency, and she wanted to do something about it. After serving as executive vp and CSO at both EntreMed and Anergen, Dr. Nacy established Sequella in 1997.
“There was an obvious market need, and we decided to create a company to take on the challenge,” says Dr. Nacy, who became the full-time CEO in 1999. Initial funding came from a stock offering and government grants. To date, the company has received more than $50 million in funding, more than half from grants. Sequella has also received 124 patents with 55 more pending.
Sequella seeks to proactively address emerging health threats, according to Dr. Nacy. Through focused execution, clear commercialization pathways, and strategic partnerships, Sequella, which has 17 employees, intends to commercialize a broad product portfolio designed to treat global health threats with significant market opportunity. After commercializing its TB products, the company plans to accelerate the development of other preclinical candidates for select indications with unmet needs.
Because some TB vaccinations can produce false positives in testing, and because faster diagnosis of TB not requiring syringes or other complicated equipment could be critical in countries where the health system is already overburdened, Sequella’s first product is a noninvasive skin patch test called the TB Patch. The transdermal patch test for TB provides a “yes” or “no” result that “looks and feels like a poison ivy reaction,” says Dr. Nacy.
Testing with the TB Patch can be done at the point of care. It requires no specimen generation and provides results within four days with 80% sensitivity and 96% specificity, according to Alan S. Klein, evp.
The Sequella TB Patch delivers MPT64, a protein specific to all organisms that cause TB. In people with active, infectious TB, a localized immune response consisting of erythema and/or vesiculation appears three to four days after application to the skin. It is easier to use and provides a shorter time to diagnosis than currently available TB tests, Dr. Nacy reports. In addition, it enables physicians to avoid wasting time, resources, and medicine treating false positive cases, she adds.
Sequella’s patch detects only active tuberculosis. Currently, initial detection of tuberculosis infection is based on a skin test that does not differentiate between patients with active infection, who are infectious, and patients with latent infection, who are not infectious. In addition, people who have been inoculated with BCG vaccine, which is still being used in many developing countries to control tuberculosis, also test positive.
The TB Patch can be marketed currently in 133 countries. The TB Patch, which is expected to be available outside the U.S. beginning in 2009, could be used in tandem with existing diagnostics to speed diagnosis of TB that has spread outside the lungs, which usually requires slow-growing bacterial cultures.
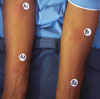
The Sequella TB Patch is a point-of-care diagnostic used to diagnose active TB infection.
Drugs in Development
Sequella demonstrated the success of public-private partnerships when it received clearance from the FDA to enter Phase I trials in 2006 with its TB drug candidate SQ109—a diamine antibiotic that could replace one or more of the current first-line anti-TB drugs to shorten or simplify therapy.
SQ109 was discovered in NIAID’s intramural laboratories in 1999, and developed with extensive support from NIAID, as well as the NCI/NIAID Inter-Institute Program for the Development of AIDS-Related Therapeutics. SQ109 was granted FDA Fast Track status, as well as FDA and European Medicines Agency Orphan Drug Designation in 2007.
An orally active tablet, SQ109 has shown in vivo activity against pulmonary TB alone and with other TB drugs, according to Dr. Nacy. With a mechanism of action distinct from other antibiotics used in TB therapy, SQ109 inhibits cell wall synthesis in a select group of microorganisms with in vitro activity against both drug-susceptible and drug-resistant TB bacteria, including XDR-TB. “SQ109 apparently makes other TB drugs work better,” she insists, “enhancing both in vitro and in vivo their activity, thereby shortening the time required to cure mice of experimental TB by 30 percent.”
SQ109 is beginning Phase Ib clinical trials in the U.S. and Phase II studies will be initiated in Africa in 2009. The drug also has in vitro activity against common nosocomial fungal infections including Candida sp. and Aspergillus sp., Dr. Nacy notes.
Another Sequella offering, SQ641, is the lead drug candidate from a 7,000-compound library of semisynthetic TL-1 inhibitors developed as potential treatments for TB or bacterial pneumonia (Streptococcus pneumoniae). TL-1 is an enzyme required for cell wall synthesis in all bacteria, including TB-causing Mycobacteria. Sequella licensed the compound library from Daiichi-Sankyo, which identified the compound class and performed extensive research and preliminary preclinical development on several drug leads. Sequella has exclusive rights to the series of TL-1 inhibitors for the treatment of TB and all other indications for nearly every worldwide market.
In September, Sequella received a $2.3 million, three-year SBIR grant from NIAID for the development of SQ641. The Phase II SBIR grant will fund the conduct of several IND-related critical path studies, including delivery optimization in vivo. According to Dr. Nacy, the drug may be both orally and parenterally active, and the company is seeking to develop it for use in the hospital and elsewhere.
“Both of these promising drug candidates would enable control of drug-sensitive and drug-resistant TB, both are active and synergistic, both would shorten the treatment time from six months to much less, and both would improve efficacy,” Dr. Nacy concludes. “These compounds have the potential to provide early and prolonged bacterial clearance during the intensive phase of TB therapy and could shorten and improve TB treatment.”







