February 15, 2010 (Vol. 30, No. 4)
Elizabeth Lipp
Scientists Explore Range of Options to Remove Roadblocks Along the New Product Development Highway
As pharma looks for better ways to build its drug pipelines, researchers are improving upon existing technologies and broadening their usage. At CHI’s recent “High Content Analysis” conference, presenters discussed new and revamped tools, and showed how these advances are helping pharma companies keep their drug pipelines active and productive.
According to Louis Stancato, Ph.D., senior research advisor, cancer growth and translational genetics at Eli Lilly & Co., phenotypic drug discovery (PDD) was previously laborious, inefficient, and often led to dead ends. “But the advent of higher volume informatics technologies, together with the high content imaging (HCI) piece, really makes PDD possible in a way that wasn’t tenable until now.”
His presentation examined how HCI subpopulation analysis tools enable a high-resolution look into cancer cell function. “This discipline is ideally suited to HCI applications. By incorporating custom in-house informatics tools, we can advance molecules with novel mechanisms of action through the lead-generation process, in particular, chemical series previously discarded owing to perceived failure in conventional targeted approaches.”
Dr. Stancato says that informatics experts help his team design custom algorithms, custom analyses, and data viewers that help find phenotypic fingerprints of interest. “Iteratively, we run our SAR looking at this phenotypic fingerprint in much the same way a chemist would look at an IC50 against an enzyme.
“We might look at upwards of 10 different data points from the same cell, synthesized to give us a number that we can then use to assess our structure activity relationships. This past summer, we launched an externally focused phenotypic drug discovery effort, called PD2 which is an open, global collaboration with academic and biotech institutions to help discover molecules.”
Dr. Stancato also examined case studies of molecules that could not have been identified any other way. They were essentially thrown away because they did not work in the targeted setting the way they were originally intended. “And if it weren’t for the imaging approach, we never would have known.”
Dr. Stancato’s group has helped many researchers with their imaging and informatics approaches by phenotypically showing molecules that were thought to behave similarly, actually behaved differently when looked at using his group’s informatics tools. “Everything we do results in a phenotypic change regardless of where it is, and that response will define whether or not the molecules we identify will help patients.”
Single-Cell Data Leveraging
Technology has indeed caught up to the ability to track phenotypic changes in cells, noted Oliver Leven, Ph.D., head of screening services, Europe, Genedata. “High-content screening experiments produce rich information on phenotypic changes of individual cells when subjected to treatment with compounds, siRNAs, or other inducers.”
“Managing resulting microscope images is a concern, but upcoming challenges to the researcher are the biologically meaningful interpretation and quantification of the high-content screening outcomes, especially with higher throughput as HCS is applied more broadly and more often.
“For example, will you be able to distinguish cell subpopulations of differential response, statistically aggregating them across cells, wells, and replicates, normalizing signals and eliminating errors, separating and quantifying phenotypes and effects for thousands of compounds?”
These challenges become exponentially significant when researchers attempt to scale up for large throughput. Dr. Leven’s presentation addressed the issue of high-content screening analysis within a high-throughput screening infrastructure, an approach that is being adopted more frequently by large pharma companies.
Dr. Leven noted that leveraging HCS data from the complex single-cell datasets—with millions of data points per plate—requires a scalable framework with automated data processing and intelligent management functions, including scientists’ review at any stage of the process.
“One point we made in the presentation is that, when using Genedata Screener for high-content screening analysis, users can reliably and efficiently create a hit list for a complex high-content screen,” said Dr. Leven. “It’s not easy to do, since there are many different features to be evaluated simultaneously, and currently, this is manually done on an ad-hoc basis without proper support. Today, however, technology is available that enables you to create a hit list with an initial rule set, run your quality control, and at any point you can go back to adapt the rules, and your changes will be reflected in the hit list—all your hit list criteria and decisions will be documented.”
JAK-STAT Pathway
Signal transducers and activators of transcription (STATs) mediate the effects of growth factors and cytokines to regulate the expression of genes involved in cell proliferation, differentiation, inflammation, migration, and apoptosis. One of several sessions that Paul A. Johnston, Ph.D., research associate professor, pharmacology and chemical biology at University of Pittsburgh School of Medicine, presided over at the meeting discussed the role of STAT3 in squamous cell carcinoma of the head and neck.
“One of my collaborators, Jennifer Grandis, Ph.D., was originally interested in developing a standard STAT3 reporter assay for screening. Reporter assays are easy to develop and fairly easy to run in high throughput, but there are also a lot of challenges associated with them post screen, there is a lot of potential for artifacts especially when you are looking for inhibitors.”
Drs. Johnston and Grandis settled on a high-content imaging approach. “I knew that there was STAT3 published procedures out there. I thought it was going to be straightforward, but it was not.”
In order to perform proof-of-principle experiments, the researchers obtained antibodies for STAT1, STAT3, and STAT5. Activated STAT3 is an oncogene that directs tumor cells toward proliferation and survival, induces angiogenesis and alters the tumor microenvironment, and promotes tumor metastases through its effects on cell migration and invasion.
In sharp contrast, activated STAT1 is considered a tumor suppressor because it is a potent inhibitor of tumor growth, promotes tumor cell apoptosis, and enhances tumor immunity. “We needed antibodies for STAT3 and STAT1 so we could look for something that selectively inhibited STAT3 without inhibiting STAT1,” said Dr. Johnston.
“We were able to test and identify antibodies that worked and were able to develop a three-colored dual-antibody assay that measures nuclei and both STAT 3 and STAT1 tyrosine phosphorylation. STAT tyrosine phosphorylation triggers STAT dimerization, nuclear translocation, DNA binding, and transcriptional activation.”
Dr. Johnston cautions that one size doesn’t necessarily fit all. “Many natural product derived anticancer compounds are not the easiest compounds to synthesize. Also, the cellular activities aren’t always restricted to a single target or pathway. I think this is the real value of content imaging.”
Small Molecule Discovery
The whole concept of flow cytometry is quite simple, observed Larry Sklar, Ph.D, principle investigator at the University of New Mexico Center for Molecular Discovery, who explained that “when people run flow cytometers, one sample is one file.
“Generally, flow cytometry doesn’t do high throughput—doesn’t do many samples very well, very quickly—so we created a platform to solve that problem. Our technology is high-throughput flow cytometry, which is intrinsically high content because you can make many measurements simultaneously.”
Eric Prossnitz, Ph.D., professor of cell biology and physiology at the University of New Mexico, spoke extensively about the HyperCyt flow cytometry platform for high-content high-throughput small molecule discovery. “The platform is evolving for 1,536-well plates and direct sample delivery. Recently, we demonstrated HTS applications with primary cells and yeast multiplex model systems for TOR pathway analysis, as well as innovative molecular assays for intracellular trafficking pathways.”
The platform can be used to make molecular measurements using assemblies of protein-protein or protein-small molecule interactions on particles. “This is a unique technology that is still in development. One of the challenges we have is keeping up with the sheer throughput of the technologies that are already well established. We can work with complex cell suspensions, bacteria, and yeast in suspension. We can look at, not just high content, but also multiple processes and multiple targets simultaneously, because of the way flow cytometry can color code samples and analyses.”
Dr. Prossnitz seeks molecules that can regulate physiological processes. “Fundamentally, we are interested in understanding complex interactions between estrogen and its multiple receptors. Until a few years ago, there were two known receptors and it was thought that all interactions were mediated by those two receptors. It’s been somewhat challenging to make headway in this space.”
“Fortunately, we isolated a compound that selectively activates the new receptor GPR30, which was published in 2006. Since then, over 30 or so publications have used this probe to ask questions in almost all areas of physiology and biology; without this probe, it would have been far more difficult to ask these questions.”
Dr. Sklar noted that the flow cytometry platform is well suited to fill a unique niche in small molecule identification for cell and molecular assays in suspension, especially in complex cell suspensions for primary cells, hematopoietic stem cells, and leukemia.
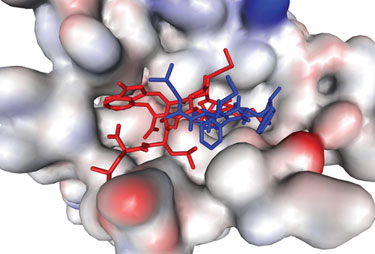
Formylated peptides (agonists=red) and phenyl-butazone derivatives (antagonists=blue) docked into a homology model of the N-formyl peptide G protein-coupled receptor
Effects of HSP90 Inhibition
Susan Lyman, Ph.D., research scientist at Exelixis, presented a cell-cycle analysis method that she and her colleagues developed. “This new method allows us to easily profile cell-cycle perturbations in a large number of samples, with improved throughput and greater information content versus classical methods of cell-cycle analysis like FACS.”
Dr. Lyman noted that cell-cycle analysis has traditionally been carried out by fluorescence activated cell sorting (FACS). The new method, in contrast, uses image-based assessment of fluorescently labeled cells, combining a measurement of DNA content with imaging of several phase-specific readouts.
“The end result is a novel and robust high-throughput Cellomics-based cell-cycle assay that reports the phase status of a cell as well as its DNA content.” Dr. Lyman and her colleagues applied this technique to examine the cell-cycle perturbations caused by inhibition of HSP90, a molecular chaperone that enhances the stability of client proteins.
“We used the high-content cell-cycle method to analyze the cell-cycle effects of several different small molecule inhibitors of HSP90 in a large panel of cancer cell lines and were amazed at how distinct HSP90 inhibitors yielded nearly identical cell-cycle phenotypes. It’s been a very useful tool for profiling small molecules,” Dr. Lyman insisted.
“In developing this cell-cycle analysis method, we tried to combine the best aspects of FACS and the best aspects of high-content imaging to generate high-quality cell-profiling data for our internal preclinical studies. Essentially, we have created a new application of some existing technologies to produce an efficient and robust means of assessing cell-cycle perturbations to help drive our preclinical development.”
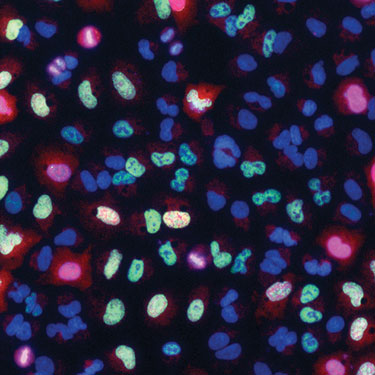
A merged multichannel image of lung cancer cells stained for DNA and for cell-cycle phase markers (Exelixis)







