February 15, 2012 (Vol. 32, No. 4)
Diana Gitig, Ph.D.
microRNAs (miRNAs) are short stretches—about 22 nucleotides long—of endogenously expressed noncoding RNA. They modulate gene expression by binding to complementary sequences in mRNAs, thereby preventing translation from them, possibly by causing their target mRNAs to be degraded. In metazoans, miRNAs usually bind to the 3´ untranslated region of their targets, and the complementarity need not be perfect; these mismatches allow one miRNA to target a number of different genes or a number of sites on the same gene.
Although miRNAs were first identified in 1993, their importance as genetic regulators was not recognized for another ten years or so. Now it is estimated that the human genome encodes on the order of 1,000 miRNAs, and their prominent role as genetic regulators means that they are important players in many essential cellular processes. They have also been found to be misregulated in many disease states.
Identifying Targets
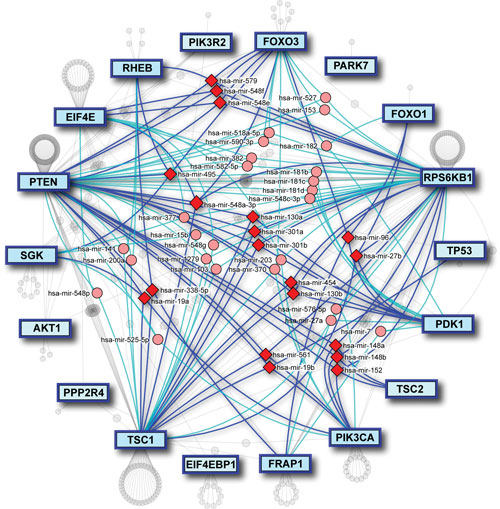
Obviously, a key to understanding how different miRNAs exert their effects depends on identifying their target genes. At least 12 different databases exist to aid in miRNA target prediction; they all use slightly different algorithms, and they all have different strengths and weaknesses.
Igor Jurisica, Ph.D., a senior scientist at the Ontario Cancer Institute, compiled all of these databases into one freely available tool called mirDIP for microRNA Data Integration Portal. He will be discussing his findings at the “microRNA International Symposium” in São Paulo, Brazil, next month.
“We can use this resource in cancer research to streamline and validate results. We can not only predict which miRNAs are relevant for a particular cancer [Dr. Jurisica is currently focusing on lung cancer], but if they up- or downregulate their target genes. Usually, when trying to implicate miRNAs in disease, people use an miRNA chip. They then have to choose which miRNAs to study and find the target genes, and that still doesn’t tell you the directionality of gene regulation.
“Our computer algorithm has all three steps in one, so you can more quickly define which qPCR experiments to do to validate the results.” Over 1,000 unique users logged on to mirDIP since it was introduced in February 2011. Using mirDIP and NAViGaTOR, a similar database compiling program that his lab generated for protein interaction networks, Dr. Jurisica categorized miRNAs into two distinct classes: universe miRNAs regulate many signaling pathways, while intra-pathway miRNAs target multiple genes within a single pathway.
mirDIP integrates 11 miRNA prediction databases, allowing users to customize their miRNA target searches. Combining miRNA predictions allows users to obtain more confidence in their miRNA targets.
Disease States and Potential Therapeutics
Youhai Chen, M.D., Ph.D., associate professor of pathology and laboratory medicine at the University of Pennsylvania, says that miR-21 is “probably the most famous microRNA there is.” miRNA-21 is an oncomiR; just like an oncogene, it is upregulated in a number of cancers and promotes uncontrolled cell growth. But Dr. Chen studies its involvement in type 1 diabetes. miR-21 targets the tumor suppressor PDCD4 (Programmed Cell Death Protein 4).
In lymphoma and leukemia, elevated levels of miR-21 promote oncogenesis by preventing the translation and accumulation of PDCD4. Dr. Chen and his colleagues have found that the same is true in pancreatic β cells, the insulin-producing cells that are killed by the immune system in type 1 diabetes: elevated levels of miR-21 suppress PDCD4, and can thus prevent the death of these pancreatic β cells.
Dr. Chen will be presenting this work at CHI’s conference on “microRNAs in Human Disease and Development” in Cambridge, MA, next month.
Matthew Evans, Ph.D., an assistant professor of microbiology at Mount Sinai School of Medicine, studies hepatitis C (HCV). This is difficult to do because the virus doesn’t replicate well in culture. Only one cell line can support it, and that cell line is far from ideal because it is not polarized like the hepatocytes HCV actually infects.
miR-122 is a liver-specific miRNA that regulates cholesterol and fatty acid biosynthesis. “Instead of inhibiting gene expression like most miRNAs, miR-122 actually promotes replication of the HCV genome by directly interacting with the far 5´ end of the viral RNA genome. This is pretty much the opposite of a normal function of a microRNA,” noted Dr. Evans, who will also be presenting at the CHI meeting.
This phenomenon was first discovered in 2005. In 2011, Dr. Evan’s lab showed that, when put into polarized HepG2 cells along with a required HCV receptor, miR-122 can support the entire viral life cycle, including infectious virion release. “This doubles the number of cell lines we can use to study HCV,” Dr. Evans says.
Interestingly, the stabilizing action of miR-122 does not require the viral RNA to be transcriptionally active, and miR-122’s normal function is not required either. Sequestering miR-122 may potentially be an effective anti-HCV therapy. miR-122 has been successfully silenced in African green monkeys, and the treatment is now in Phase II trials.
Just down the hall from Dr. Evans, classically trained virologist Benjamin tenOever, Ph.D., set out to determine if miRNAs play a role in the cellular response to viral infection. Alas, they do not.
But Dr. tenOever persevered; he figured, “Just because miRNAs aren’t antiviral, doesn’t mean they couldn’t be!” His lab then generated a flu vaccine by grafting an miRNA target site into the influenza virus. He chose an miRNA that is present in mammalian but not chicken cells, so the virus grows perfectly well in eggs and is only attenuated when it infected mammalian cells that endogenously express the miRNA. Unlike canonical miRNA target sites, which are not perfectly complementary to the miRNA, Dr. tenOever made this target perfectly complementary.
The miRNA thus acted more like an siRNA, attenuating viral replication 1,000-fold instead of the two- to threefold usually achieved by miRNAs. And unlike other flu vaccines, this one does not rely on the immune system to destroy it, so it can be administered to immunocompromised individuals without eliciting unpleasant side effects.
Dr. tenOever has played with engineering miRNAs and their target sites into other viruses as well, generating viral vectors targeted to specific cell types—much like HCV evolved to replicate only in hepatocytes because of its reliance on miR-122, which is expressed only in those cells.
He has also made viruses that can churn out an miRNA of choice once inside cells. “We don’t work with viruses and miRNAs in a conventional sense,” says Dr. tenOever, “but we do a lot of things with them, and our toolbox for molecular therapeutics has grown because of it.”
Convergence and Divergence
Similar to the way Dr. Jurisica distinguishes between universe and intra-pathway miRNAs, Walter Lukiw, Ph.D., associate professor of neuroscience, ophthalmology, and genetics at Louisiana State University Health Sciences Center, talks about miRNA convergence and divergence. He highlights that multiple miRNAs can converge onto a single mRNA, and a single miRNA can diverge to regulate the expression of multiple messages.
“This is one strategy that nature uses to control gene expression,” notes Dr. Lukiw, another CHI conference presenter.
The idea is near to his heart, as his laboratory identified the first example of miRNA convergence: miR-125b, miR-146a, and miR-155 all suppress the transcription of the complement factor H (CFH), a protein involved in innate immunity and the inflammatory response.
Both miR-125b and miR-146a are upregulated in Alzheimer disease, and each has other targets in addition to CFH. Among them, miR-125b targets the cell cycle inhibitor CDKN2A to promote glial cell proliferation, a hallmark of neurodegeneration, and miR-146 targets Interleukin-1 Receptor Associated Kinase 1 (IRAK) 1 and 2 to promote neuroinflammation. Thus the elevated levels of these two miRNAs can account for a number of the key pathological features that define Alzheimer disease.
“Many disease processes—Alzheimer, Parkinson, ALS, Down syndrome, schizophrenia—are regulated by multiple mRNAs,” says Dr. Lukiw. “If one or a few miRNAs can control all of the messages involved, an antisense strategy targeting that miRNA could knock down a whole family of pathogenic genes.”