June 1, 2006 (Vol. 26, No. 11)
Turning Individual Wells from Microtiter Plates into High-Quality Mini-Reactors
Construct and mutant libraries, as well as collections of bacteria, fungi, and streptomycetes and eukaryotic cell lines are being used in a wide range of research areas. As diverse as these areas may beranging from traditional streptomycete screening aimed at the discovery of new drugs to the use of RNAi libraries in recent yearssmaller culture volumes are the common denominator.
Receptor-binding assays, cell-based inhibition assays, and other bioassays are being miniaturized to an extent that they can be performed with pg or ng amounts of cell products. The same holds for LC-MS assays for biocatalyst screenings and medium optimization studies.
The challenge of the last five years has been to miniaturize microbial and cell growth systems while maintaining the growth and axenic qualities achieved in shake flasks and bioreactors. The use of the standard microtiter plate format is attractive because of the availability of compatible robots, readers, and other equipment. Methods to turn individual wells from microtiter plates into mini-reactors are described in this tutorial.
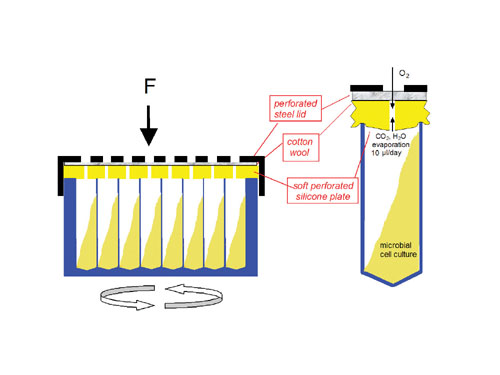
Fig 1: Schematic view of a sandwich cover for deep-well plate cultures to reduce evaporation and allow vigorous orbital shaking without cross contamination
Well Closures
The most critical aspect of microbial growth in microtiter plates is the closure system of the individual wells, which has to prevent cross-contamination (even during vigorous shaking), permit the exchange of headspace air, and limit evaporation.
Equal physical conditions in all wells is a further requirement: the wells in the corners should have exactly the same characteristics as the wells in the middle of the microtiter plate. This is especially important in the area of medium optimization and mutant screening where productivity improvements as low as 5% should be detectable.
Gas permeable membrane filters (sealing tapes) are available from many suppliers, and are well-suited for qualitative work, but have a number of distinct disadvantages for quantitative work. First, during vigorous shaking, droplets may cover smaller or larger parts of the membrane, leading to random well-to-well variations in evaporation and supply of oxygen. Second, the partial dissolving of potentially toxic glue material may harm growth.
An additional drawback of plastic membranes is their relatively low diffusion coefficient for oxygen and high diffusing coefficient for water. The result is substantial water loss by evaporation while the oxygen demand of the culture is often not met.
A solution that overcomes these problems with sealing tapes has been developed by Enzyscreen (), is manufactured by Kuhner (www.kuhner.com), and distributed in the U.S. by Biopro International (www.biopro.com). The solution consists of a sandwich cover with three layers: a silicone layer with 96 small holes in the middle of each well, a layer of cotton wool, and a stainless steel lid with 96 holes (Figures 1 and 2). The sandwich cover and microtiter plate are clamped together tightly in order to prevent spillage of the culture fluid during high G-force orbital shaking and to ensure that exchange of headspace air occurs solely through the center holes.
In this system, the rate of exchange of headspace air and evaporation is controlled by the diameter of the holes in the silicone layer; holes of 0.5𔂿 mm result in water evaporation rates of 10󈞀 µL per well per day at 50% humidity and 30C.
A thin stainless steel foil with 200-µm holes inserted between the silicone layer and the cotton wool layer results in even lower evaporation rates (4𔃄 µL per well per day) and is advisable for 100�-µL cultures in low-well plates. Such evaporation rates correspond to 2𔃃% of the culture volume per day, which is acceptable for most purposes. Therefore, no air humidification is required, which avoids the nuisance of fungal wall growth, often seen in humidified incubators.
Since the diffusion coefficients of oxygen and water in air are practically equal (0.21 cm2/s at 20C), it can be readily calculated that an evaporation rate of 10 µl per day at a relative humidity of 50% correlates with an exchange rate of headspace air of 450 µl (containing 3.6 µmol O2) per minute, provided a mechanical diffusion barrier such as cotton wool is used. This guarantees a sufficient oxygen concentration (above 18%) in the headspace even at oxygen consumption rates in the culture as high as 40 mmol/L/h. Air supply rates in this range correlate well with the general rule of thumb for lab-scale stirred tank bioreactors: one vessel volume of air per minute.
A sufficiently high and universal exchange rate of headspace air is a prerequisite, but not a guarantee, that the cells growing in the wells are supplied with enough oxygen. Gas-liquid transfer is another limiting factor.

Fig 2: Sandwich covers convert all individual wells of square deep-well plates into 96 minireactors
Orbital Shaking Conditions
The type of microtiter plate to be used is project-dependent. For culture volumes of ~250 µL, good results can be obtained with regular polystyrene 96-low-well plates, as long as one is prepared to settle for moderate oxygen transfer rates.
One advantage of low-well plates is that they can be stacked, provided special sandwich covers are used that maintain a space between the cover and the microtiter plate put on top (Figure 3). Shaking at 300 rpm and a 50-mm shaking amplitude results in oxygen-transfer rates (OTR) of around ~15 mmol O2/L/h.
This OTR can be almost doubled by lowering the culture volume to 100 µL or by increasing the orbital shaking frequency to 350 rpm. The hydrodynamic pattern during shaking is regular and nonturbulent, so it is also suitable for strains and cell cultures that are susceptible to shear forces.
For higher OTRs (up to 40 mmol/L/h) and culture volumes (0.5?? mL), the use of square 96 deep-well plates is advisable. The square shape in the horizontal plane results in a turbulent shaking pattern (Figure 4) and therefore better mimics the situation in stirred tank bioreactors than roundwell microtiter plates do.
For this reason, square deep-well plates are especially well-suited for medium optimization studies for large-scale production of secondary metabolites. A major determinant of good aeration rates is the shaking amplitude. The best results are obtained by relatively high shaking amplitudes (50 mm or more) since the generated G-forces are sufficiently high to induce mixing to the bottom of the wells. Cell densities of almost 10 g dry wt per liter are attained at growth rates similar to shake flasks incubated under optimal conditions (Figure 5).
Square 24-well deep-well plates at culture volumes of 2?? mL are favored in some research areas, such as medium optimization for streptomycetes or fungi. A major reason for the use of 24-well plates for pellet-forming microorganisms is to reduce the effect of statistical variations in the number of pellets per well on the concentration of the metabolite. It should be noted that for 24-square well plates, 300 rpm at an amplitude of 25 mm, instead of 50 mm, is sufficient to achieve aeration rates of 40?? mmol/L/h.

Figure 3. Low-well microtiter plates can be stacked on an orbital shaker if special sandwich covers allowing a free airflow between the plates are used.
Other Aeration Methods
The use of orbital shaking is attractive because it is effective, economical, and simple. Furthermore, it helps to minimize well-to-well variations since the G-force is identical in each well and therefore the hydrodynamic patterns and oxygen transfer rates show little variation. The use of small stirring bars or air-bubbling systems is more elaborate and almost inevitably leads to higher well-to-well variations.

Fig 4: Shaking patterns during orbital shaking of a 96 square deep well at 300 rpm
Inoculation of Wells
For some strains, such as E. coli, it is possible to inoculate microtiter plate cultures from a thawed master plate using a pipetting robot. A distinct disadvantage of this procedure, however, is that for most micro-organisms, 90󈟏.9% of the cells remaining in the master plate lose viability with each freeze-thaw cycle. For other heterogeneous culture collections, an additional drawback is that if cells from frozen stocks are inoculated directly into liquid cultures, the viability is often quite poor.
Instead the following procedure is recommended. The strains are stored in the presence of 15% (v/v) glycerol in deep-well microtiter plates at -80°C. A special, sterile, replicator (commercially available by Enzyscreen and Khner) with 96 spring-loaded pins is pressed onto the 96 frozen glycerol stocks (80°C), and samples 0.3 µL per well by heat transfer, without thawing the remaining glycerol culture (Figure 6).
Since the master plate remains frozen, the viability is not affected by repeated sampling. The same master plate can be used hundreds of times. The sampled cells are transferred onto an agar medium suspended into a microtiter plate. The resulting colonies can be used after 1𔃁 days for inoculation of the liquid cultures.
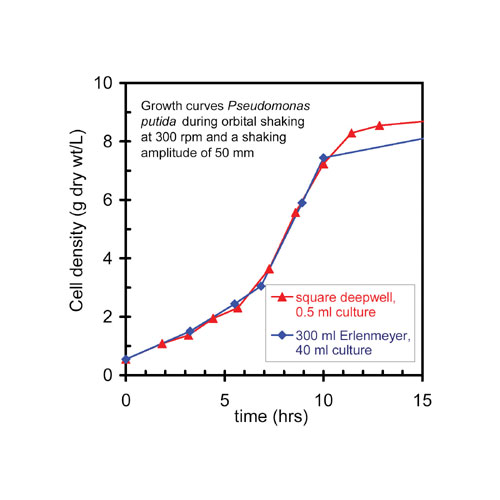
Fig 5: Growth curves of a Pseudomonas putida strain in wells of a deep-well microtiter plate and an Erlenmeyer shake flask during orbital shaking at 300 rpm and at 300 rpm and a shaking amplitude of 50 mm
Miniaturization in the Future
For suspended cultures, an intrinsic hurdle for further miniaturization is the surface tension and cohesion of aqueous cell suspensions. In culture vessels smaller than ~3.5 mm in diameter, the surface tension cannot be overcome by the centrifugal forces applied by orbital shaking. The result is a dramatically low OTR and absence of any significant mixing of the liquid, even if G-forces above five are applied. These intrinsic physico-chemical limitations have led us to believe that the scope for high-quality cultivation of liquid cultures at this scale (e.g., in 384-well plates) is minimal.
Further parallelization (more cultures per square meter) is more likely to be achieved by reducing the height of the cultures and stacking them.
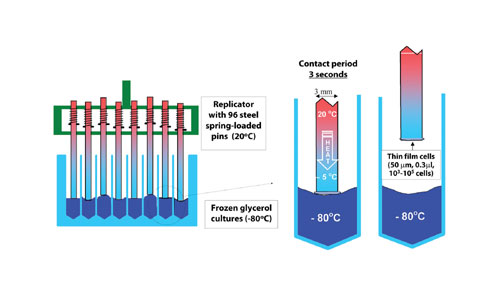
Fig 6: Sampling of 96 frozen microbial stocks without thawing the remaining culture by using a spring-loaded replicator







