March 15, 2010 (Vol. 30, No. 6)
Deepak Sharma Ph.D.
Advances Allow Researchers to Compare Images Taken at Different Times and Settings
Fluorescence has emerged as a widely used signal in life science research. In particular, fluorescence microscopy techniques have enabled researchers to characterize molecular interactions in natural cell environments. These techniques have been enhanced by electron multiplying charge coupled device (EMCCD) cameras providing sub-electron read noise and enabling visualization of everything from single molecules and sub-cellular structures to whole animals in vivo.
Fluorescence microscopy has advanced all stages of the drug discovery process, including target discovery, candidate screening with cell-based assays, toxicology evaluations, and mode-of-action studies. However, analyzing the wealth of knowledge from imaging experiments can be difficult. For years, life science researchers have struggled with the irreproducibility of images from CCD and EMCCD cameras. The analog-to-digital units (ADUs) in which those cameras report imaging data are known by many names, including gray-scale units and fluorescence units.
The problem is that ADUs are merely electronic representations of the number of photons hitting the sensor. The way in which cameras make this representation varies, even between the same make and model of camera. So an ADU doesn’t directly correlate with real incident light.
While technological advances have brought quantitative elements to imaging—such as cell counting and structural measurement—solutions for quantitation of fluorescence intensity, independent of users and equipment, is far from standard.
This is a hurdle for scientific data sharing, especially for multisite collaborations. The variable nature of the raw ADUs that cameras produce limit the labs’ ability to control for experimental variability and directly compare imaging data. Thus, knowledge produced by imaging experiments can be difficult to interpret.
Why ADUs Are Arbitrary
When scientific images are acquired on a CCD or EMCCD camera, the observed pixel value is impacted by many factors, including variable gain settings, sensor aging (in the case of some EMCCD cameras), and camera-to-camera differences in sensitivity and bias (or offset) settings.
Essential for visualizing dim signals from live cells is the high signal-to-noise ratio provided by EMCCD cameras. Those cameras multiply photoelectrons generated by incoming photons into many more electrons by moving the charge through a high-voltage extended register—the effect of which is called EM gain. However, actual applied gain can often diverge from the indicated multiplication selected by the user.
Manufacturers most often do not report exact values, so a requested gain factor by a user may in reality be slightly different from what a camera provides. For some older camera designs, the applied multiplication also might not scale linearly, such that a gain setting of 800 does not actually produce twice the signal as that of 400. This all means that the gray levels assigned in the produced image can diverge from the actual incoming photons that they represent.
Some EMCCD chips are known to “age”. Their actual applied EM gain reduces over time as the device is used. The camera that once had a true measured EM gain of 400 will not have the same gain when put at the same setting as weeks pass. Thus, an equivalent number of photons can produce a different observed response in ADUs as the camera is utilized and time passes. When not corrected for, these variables confine the meaning of an ADU to a single camera’s behavior at a single time point.
Controlling Errors
Controlling the unknowns introduced by the imaging system itself has been key to the emergence of high-content, high-throughput screening platforms, which use sophisticated hardware calibration and software to report data in a standardized manner.
While secondary screening and lead optimization can be enhanced by automated microscopy methods, not all experiments are so amenable. These experiments can be hard to integrate into the discovery process because imaging data is reported in units of ambiguous meaning and internal controls must be set up to account for such potential issues.
Even when using such controls, data represented in ADUs leaves researchers without the means to verify that controls between experiments actually behaved equivalently, unless performed in the exact same system and, in the case of systems containing aging EMCCD sensors, around the same time. Without such affirmation, integrating data from these experiments and confidently reaching statistically significant conclusions is difficult.
For example, antibodies can introduce experimental variability. They can degrade during the course of multiple imaging experiments. This results in a reduction of their binding efficacy, changing the behavior of the fluorescent reporting system. The slow degradation of an antibody would gradually change the property of the experiment over time.
Cells transfected with fluorescent proteins are also useful for many types of studies. However, transfection efficiencies often vary from cell to cell. Under- or overexpression of fluorescent proteins can introduce experimental artifacts, change molecular behaviors, and modify intracellular distribution. Likewise, fluorescent dye uptake can also vary from cell to cell.
Without accounting for variation between the controls of different experiments, conclusions drawn from comparing such experiments have the potential for introducing additional variables and resulting in further errors. Such errors could potentially influence outcomes of the study such as the selection of candidate drug compounds.
Reporting imaging data in absolute, quantitative units lays the foundation for individual researchers and collaborators across the globe to more completely control for experimental variation. This makes imaging experiments reproducible and more easily comparable between different imaging systems, personnel, and laboratories.
Of course, researchers can manually calibrate their current cameras to align reported ADUs with actual incident photons. However, the calibration and back-calculation process requires a high level of knowledge, specialized equipment, and staff time. Since the process must be repeated regularly to account for sensor and gain aging of some EMCCD devices, it can be extremely cumbersome for researchers or technicians to keep every microscope camera characterized.
Implementing standards for reporting imaging data, starting with an absolute unit of measurement, will increase the potential for reporting statistically significant results and boost confidence in the reliability of those results.
Photons that hit CCD and EMCCD sensors generate photoelectrons, the fundamental unit of measurement for these devices. The way those photoelectrons are processed into ADUs is done in a variable manner, dependent on the camera electronics and the sensor.
Imaging data reported in photoelectrons, which directly reflects incident photons, would provide an objective unit of measure independent of instrument and researcher or camera-specific processing. A novel re-engineering of camera technology has enabled researchers to use the photoelectron as the standard measurement in imaging experiments without the need for manual characterization, technology expertise, back calculations, constant maintenance, or additional equipment.
Evolve EMCCD cameras from Photometrics perform the necessary characterizations, calibrations, calculations, and real-time measurements to determine the exact conversion factor and bias (offset) values of the camera—allowing it to report measurements in photoelectrons (Figure).
By standardizing the unit of measurement, Photometrics’ technology allows researchers to compare, in absolute terms, images taken at different times and gain settings.
Transitioning fluorescence microscopy from a qualitative to a quantitative technology not only advances academic research, but also enables a pharmaceutical company’s diverse network of researchers and laboratories to maintain data integrity over the duration of the drug development cycle—independent of researcher and equipment turnover.
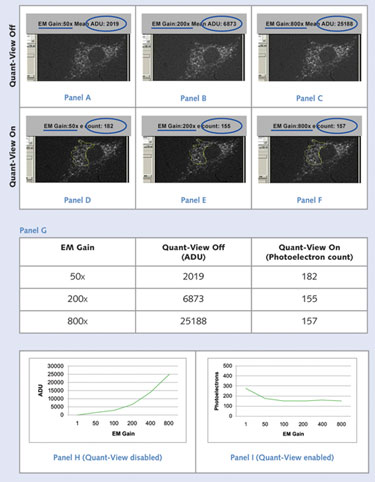
Panels A to F: A low-light cell sample with stained Golgi apparatus; Panel G: Average pixel data from Panels A to F; Panels H and I: Average pixel data from images taken at six different EM gain settings
Deepak Sharma, Ph.D. ([email protected]), is senior product manager at Photometrics. Web: www.photometrics.com.