January 1, 1970 (Vol. , No. )
Cathy Vaillancourt, Ph.D. INRS
Sean Taylor, Ph.D. Bio-Rad Laboratories
The MIQE guidelines and papers such as the one discussed here can be applied to avoid pitfalls.
Reverse-transcription quantitative polymerase chain reaction (RT-qPCR) is everywhere. Its ubiquity, however, does not mean its practitioners produce consistent and reproducible results that reflect the underlying biology. The exquisite sensitivity of RT-qPCR demands rigorous experimental design; the Minimum Information for Publication of Quantitative Real-Time PCR Experiments (MIQE) guidelines were published to help standardize this technique 1, 2. Although a growing number of labs are applying the MIQE criteria to their experiments, many groups still ignore them, thus increasing the potential of publishing artifactual and inaccurate data. In fact, the erroneous conclusions from previous studies have not only misdirected research but have also had poignant societal impact [for example, see 3, 4].
RT-qPCR employs four principal steps:
- Sample extraction from cells or tissue in a rigorous and reproducible manner;
- RNA isolation and purification;
- Reverse transcription to convert the mRNA to cDNA;
- The qPCR reaction to quantify levels of reverse transcribed cDNA in the sample.
Even in the hands of an experienced researcher, any of these steps can be improperly executed resulting in data where the associated interpretations and conclusions do not reflect the biological outcome. This has been shown recently with both human placental and breast cancer studies where even opposite conclusions could be drawn from the data that was entirely due to improper sample validation and reference gene selection 5, 6. The application of a few simple and fast validation tests can assure that the final RT-qPCR data are solid 7.
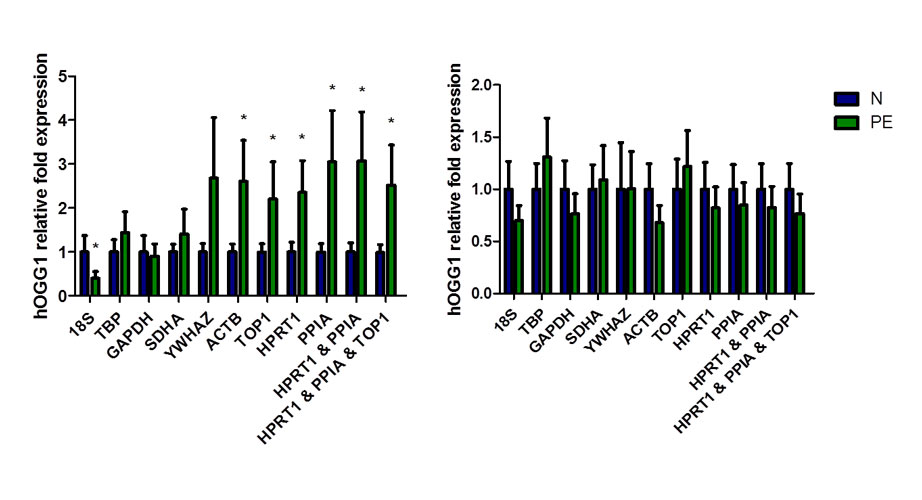
In the human placenta for example, nine candidate reference gene targets were tested for expression stability including 18S rRNA, GADPH, and beta-actin in pathological (e.g., preeclampsia) pregnancy conditions using the software tools geNorm and NormFinder. The normalized expression data for hOGG1 gave the expected or opposite conclusions when normalized to the most or least stable reference gene targets, respectively (Figure 1A) 6. Furthermore, although many labs test their RNA samples for purity (OD 260/280), much fewer test for quality—the level of degradation—which can skew the results between sample groups leading to erroneous conclusions (Figure 1B). Since about 20% of placenta tissue RNA samples can be significantly degraded 6, it is important to test sample quality. Quality can be assessed using the RQI output from Bio-Rad’s Experion Automated Electrophoresis Station 6.
The desire to turn around RT-qPCR data quickly and/or ignorance associated with the historical biases of the protocols passed down and shared between or within labs has led to a potential lack of experimental rigor. As a result, many articles published today may have inaccurate results with misleading conclusions. The MIQE guidelines and papers such as the one discussed here can be easily applied to avoid these pitfalls and strengthen the overall quality of published data.
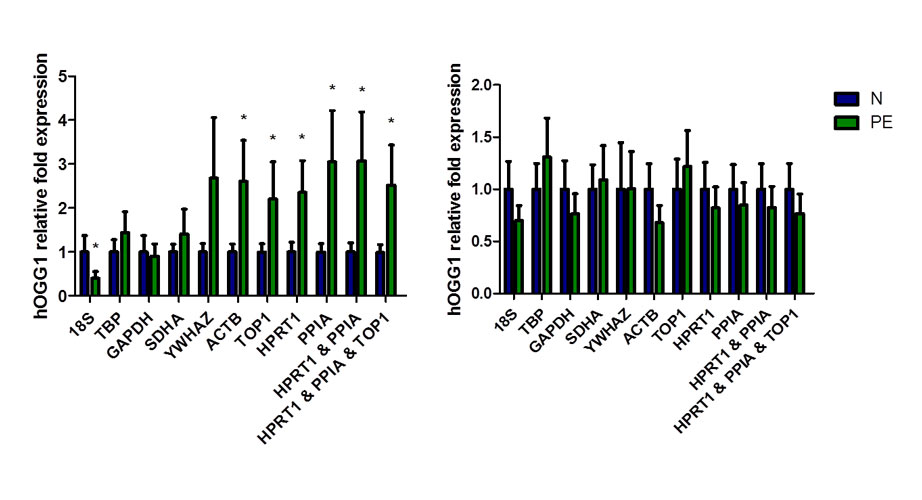
Figure 1: Effect of reference gene normalization and RNA degradation on hOGG1 (human 8-oxoguanine glycosylase) expression in placentas from normotensive (blue) and preeclamptic (green) pregnancies. (A) Variability of hOGG1 expression normalized against the tested reference genes and the best combinations (n = 11). (B) Effect of RNA degradation, three degraded RNA samples from normotensive and preeclamptic pregnancies placentas were pooled with 11 intact RNA samples of each group respectively. Mean ± SD; * P < 0.05.
Cathy Vaillancourt, Ph.D., is a professor at INRS-Institut Armand Frappier. Email: [email protected]. Sean Taylor, Ph.D., is a field application specialist at Bio-Rad Laboratories. Email: [email protected].
References:
1 Bustin, S. A., Benes, V., Garson, J. A., Hellemans, J., Huggett, J., Kubista, M., Mueller, R., Nolan, T., Pfaffl, M.W., Shipley, G.L., Vandesompele, J., and Wittwer, C.T. (2009). The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clinical Chemistry, 55(4), 611–622.
2 Bustin, S. A. (2010). Why the need for qPCR publication guidelines? The case for MIQE. Methods, 50(4), 217–226.
3 Bohlenius, H., Eriksson, S., Parcy, F., and Nilsson, O. (2007). Retraction. Science, 316 (5823), 367.
4 The Editors of Lancet. (2010). Retraction—Ileal-lymphoid-nodular hyperplasia, non specific colitis, and pervasive developmental disorder in children. Lancet, 375 (9713), 445.
5 Taylor, S., Buchanan, M., and Basik, M. (2011) A MIQE Case Study—Effect of RNA Sample Quality and Reference Gene Stability on Gene Expression Data. Bio-Rad tech note 6245.
6 Lanoix, D., Lacasse, A-A, St-Pierre, J., Taylor, SC, Ethier-Chiasson, M., Lafond, J. and Vaillancourt, C. (2012) Quantitative PCR Pitfalls: The Case of the Human Placenta. Molecular Biotechnology, Online First™, 17 April 2012, DOI 10.1007/s12033-012-9539-2
7 Taylor, S., Wakem, M., Dijkman, G., Alsarraj, M., and Nguyen, M. (2010). A practical approach to RT-qPCR-Publishing data that conform to the MIQE guidelines. Methods, 50 (4), S1–S5.