September 1, 2011 (Vol. 31, No. 15)
Jonathan Witonsky
Increased Focus on Difficult-to-Transfect Cells Driving a Burgeoning Market
In 2010, an estimated 180 million transfection reactions (not counting transfections using viral delivery tools) were conducted at about 22,500 laboratories around the world.
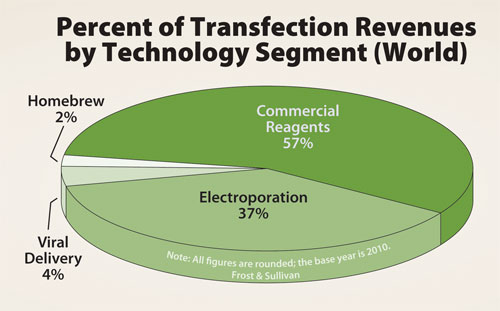
The majority of these reactions—roughly 55%—were conducted using commercial transfection reagents: kit-based cationic lipids, dendrites, or other chemicals from vendors such as Life Technologies, Roche Applied Science, and Qiagen. Electroporation is the next most widely employed transfection technology, used for approximately 25% of transfection reactions. Finally, homebrew reagents were used for the remaining 20% of transfection reagents.
The global sales of transfection products generated approximately $220 million in revenue in 2010, and the market is forecasted to grow at a compound annual growth rate (CAGR) in the region of 8% from 2011 to 2016, reaching an estimated $350 million in revenue in 2016.
While commercial transfection reagents are employed for the majority of reactions, the global transfection market is undergoing a major shift; electroporation is overtaking commercial reagents as the primary driver of market growth.
As recently as five years ago, commercial transfection reagents were outgrowing electroporation. In 2005, for example, the commercialization of optimized reagents for the transfection of siRNA allowed for market growth to exceed 20%. Despite continued double-digit growth in RNAi and protein production applications, the market for commercial transfection reagents is expected to grow at roughly 7% CAGR from 2010 to 2016.
Today, the demand for solutions that enable efficient transfection in primary cells is resulting in an electroporation market that is forecasted to grow with more than a 10% CAGR between 2010 and 2016. Electroporation market growth is attributable to biomedical researchers’ increasing average investment per reaction, which is forecasted to grow from approximately $1.70 in 2010 to $2.45 in 2016.
In 2010, approximately 1.7 million electroporation-based transfections cost more than $10 per reaction. In 2016, more than 4.9 million electroporation-based transfections are likely to cost more than $10 per reaction.
Despite high growth of primary cell transfections, the majority of transfections are conducted using cell lines. Transfections of the top five cells lines, including 293, CHO, HeLa, 3T3, and COS cells, make up as much as 50% of the total number of transfections.
Low-priced solutions, such as commercial transfection reagents or homebrew transfection reagents, provide sufficient results when transfecting these cell lines. For these applications, the higher efficiencies or higher yields allowed by costly transfection solutions are unnecessary, and therefore, customers will continue to leverage the lowest-priced products and homebrew methods.
Additionally, the use of homebrew reagents for transfection remains in practice within many laboratories. An estimated 20% of transfection reactions are conducted using homebrew methods. The widely available and inexpensive calcium chloride dehydrate, sodium phosphate dibasic, and HEPES buffer may be used to transfect without the purchase of a commercial reagent.
Price is the single most competitive advantage of using these chemicals for transfection. Homebrew methods require more manual steps, are more susceptible to human error, generally take longer, can be toxic, are not standardized, and lack the reproducibility of off-the-shelf products. However, the low price of these chemicals compared to commercial reagents has kept these methods in practice.
Academic laboratories with budgetary restraints and inexpensive labor will likely continue to use homebrew methods until funding improves or until they switch their focus to cells that are too difficult to transfect using homebrew methods. Other researchers may opt to use homebrew chemicals and commercial reagents interchangeably, depending on the cell type or the quality needed for a particular downstream application.
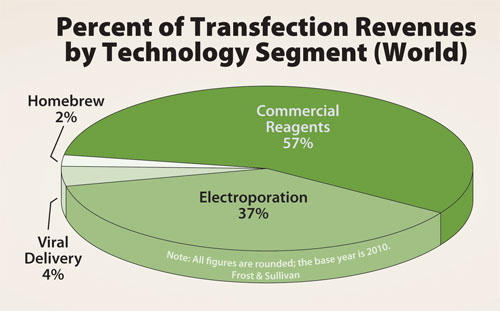
Although commercial transfection reagents are employed for the majority of reactions, electroporation is slowly overtaking them as the primary driver of market growth.
Jonathan Witonsky ([email protected]) is a principal analyst in the life sciences group of Frost & Sullivan.