November 1, 2013 (Vol. 33, No. 19)
Kathy Liszewski
Choosing the optimal time to develop a companion diagnostic alongside therapeutic products can feel like aiming at a moving target. To hit the bull’s eye, one must select the appropriate assay platform for the early validation of predictive biomarkers.
This requirement and several related challenges will be discussed at CHI’s conference on “Clinical Biomarker Assay Development.” Besides taking up the issue of choice of assay platform, presenters will discuss analytical validation strategies, reimbursement requirements, and regulatory compliance issues.
Many pharmaceutical and biotechnology companies employ biomarker analysis for pharmacokinetic/pharmacodynamic (PK/PD) modeling. Often, companies do such evaluation utilizing commercially available assay kits. “The problem is, whether they use home-brewed or commercial kits, groups must be aware of and plan for controlling lot-to-lot kit or assay component variability. This is critical for accurate bioanalysis in support of both preclinical and clinical studies,” says Afshin Safavi, Ph.D., svp of bioanalytical operations and founder, BioAgilytix Labs.
According to Dr. Safavi, even if groups have a validated assay, it is important to establish processes for sample analysis that minimize the effects from the kit and/or reagent variability to see the drug effectiveness. “When assessing the impact of a drug, if you are looking for 10–20% change in the biomarker, then a higher degree of assay rigor is required as opposed to expecting to see a three- to fourfold difference.”
Controlling lot-to-lot variability is even more critical when dealing with multiyear studies. “The concept of applying correction factors between different lots of assay kits should be discussed and planned before the initiation of the project to generate quality data so the clinicians and statisticians can make the correct conclusions and decisions.”
Since there is no standard for such control factors, each lab must set up its own quality control strategy. “Labs need to check the expiration dates of kits and have enough for studies that are expected beyond that date. At BioAgilytix Labs, we anticipate more than three months prior to changing kits or to kit expirations. We have conducted studies lasting more than three years and have found that even the same manufacturers can have lot-to-lot variations greater than 200%.”
Dr. Safavi says the bottom line is the need for communication among scientists, clinicians, and statisticians on a project. “Groups need to spend more time in development to work out the kinks. If you do not have a robust assay, the project will suffer later.”

BioAgilytix Labs advocates communication among scientists, clinicians, and statisticians to control for variability of commercial kits and assay components.
Fit-for-Purpose Assays
In drug development, biomarker assays may serve purposes other than the ones they serve in clinical studies. This possibility has been explored by John L. Allinson, vp of biomarker laboratory services at ICON’s development solutions service division. “While diagnostic assays are used for the purpose of routine safety or efficacy assessments (under recognized global clinical laboratory accreditation schemes), there are many other uses that their versatility lends them to,” says Allinson. “These include, but are not limited to, drug candidate attrition and refinement, protocol design, dose selection, PK/PD modeling, patient stratification, companion diagnostics, post-approval surveillance, and market differentiation.
“With such a wide range of applications, often the biomarker assay may not require as much development or long-term robustness (sometimes known as ‘exploratory’ markers and assays), while other uses may actually require more intensive attention to things like precision in parts of the analytical range not associated with significant diagnostic assessment but where more subtle changes need to be characterized, for instance, due to pharmacodynamic effects of specific drugs.”
Allinson says that, therefore, in drug development biomarker assays, more or less attention can be applied to certain assay performance characteristics such as precision and limits of quantification. This leads to a tiered approach to assay validation in drug development assays versus a very well-established and standardized approach to diagnostic assays, which is required for their accreditation.
Biomarker assays are not usually off-the-shelf services. “To be absolutely certain that assays are fit-for-purpose, that is, appropriate for their intended use, there must be people in the service provision who understand the assay’s clinical utility. Matching assay and technology choice to attain a suitable assay performance that enables the correct interpretation of the results to be made is absolutely critical to providing a suitable biomarker assay service. Once these decisions are made, applying a suitable fit-for-purpose validation approach to each assay is the next critical component.”
When these careful and methodical approaches are not followed, trouble can ensue. “During my 20 years in contract research, I personally acquired knowledge of the many ways laboratories may produce data that leads to incorrect clinical decisions,” recalls Allinson. “Laboratories may modify diagnostic assays without understanding how those assays were developed.
“An example is the replacement of calibration reference material with substances that are not recognized international reference materials. If the substances behave differently from the appropriate reference material, they can give rise to different concentration data. Depending on the use of the results, erroneous data can have very serious consequences.”
Precision Medicine
The pharmaceutical and diagnostic industries are rapidly moving toward a more proactive precision medicine approach that stratifies patient populations. “Precision medicine differs from personalized medicine,” notes Shane Weber, Ph.D., director of diagnostics, clinical research, and precision medicine at Pfizer.
“Personalized medicine seeks to help each individual patient, such as when creating a personal vaccine for a cancer patient. On the other hand, precision medicine is an approach that stratifies and segments patients into highly responding groups. An example would be KRAS mutations that underlie a variety of cancers. The use of biomarkers, diagnostics, and endo-phenotype stratification strategies are opening the door to faster and less expensive precision medicine clinical trials that will lead to therapies dramatically benefiting patients.”
Dr. Weber says that a key question is whether early biomarker assay verification needs to be done during upstream development or in parallel with companion diagnostic clinical validation. “The answer is that both are valid. Sometimes it is possible to stratify patients early into segments. At other times it is a catch-up game—when patient trial studies reveal subsections that unexpectedly responded extremely well, while most others in the population did not. The issue is that the FDA wants biomarker verification in Phase IIa and IIb and companion diagnostic validation in Phase III.”
The importance of correctly navigating regulatory compliance with the FDA is critical. “For example, under the FDA’s investigational device exemption process, the biomarker assay analytical verification process requires that the assay in U.S. clinical trials comply with use as an investigational use only (IUO) assay within a CLIA laboratory.”
Ultimately, there are three major considerations to the successful development of a companion diagnostic. “The first is to coordinate with various groups at your institution to interact on the project, Dr. Weber says. “At Pfizer, for example, our biobank and diagnostic groups frequently need to interact.”
“A second consideration is partnering. We partner outside the company with diagnostic companies that have the appropriate technology platform for the given biomarker and preferably a global commercial medical system footprint. Third, as the clinical data is generated, we continue to modify and/or stratify patient segments within the constraints of the clinical trial process in order to keep the program moving along so as to benefit patients as soon as possible.”
Diagnostic Platform
The FDA defines a companion diagnostic device as one that provides information that is “essential for the safe and effective use of a corresponding therapeutic product.” When selecting the best platform for validating a companion diagnostic, one must weigh several key considerations. In addition, “…the approach for the development and approval process is dependent on each specific program,” says Ron Mazumder, Ph.D., global head of research and product development at Janssen Diagnostics. “This means that the type of biomarker and selected assay must be considered for each individual approach.”
One key factor is assay sensitivity. “It is important to choose an assay that reflects the needed level of detection. A number of assays may show concordance, but one may be better than the other, such as TaqMan, mass spectrometry, or next-generation sequencing (NGS).”
Another important factor is assay development. “From a regulatory perspective, scientists must develop an assay that the FDA will approve and thus must know the newest regulatory guidelines. If assays have poor design style or manufacturing standards, it will be hard to get platform approval.”
Throughput is an additional consideration. “This really depends on the patient population. Often therapeutics and companion diagnostics may be for specially targeted patient populations, so throughput here is not critical. But if you have 100,000 patients and 10 sites that are involved, then you must consider the optimal platform for this level of throughput.”
But even the best platform requires something else—accessibility. “You need to select a platform that has unlimited access. New technologies such as NGS can be very useful, but at the moment, NGS is not widely available. While today it is largely used in research, in the near future, NGS will become much more user-friendly and commonly employed.”
Which Assay to Use?
Almost every new oncology drug being developed today is targeted toward a specific protein or family of proteins. Thus, companion diagnostic assays are needed to help identify those patients who will benefit from a new drug. There are a number of methods and platforms for which the companion diagnostics can be applied. Dako, an Agilent Technologies company, focuses on immunohistochemistry (IHC), fluorescent in situ hybridization (FISH), and bright-field chromogenic in situ hybridization (CISH) assays on fully automated systems.
According to Hans Christian Pedersen, principal scientist at Dako, “A companion diagnostic needs to be available wherever the drug is intended to be used. Thus, the selected technology must be adaptable into routine clinical practice and the costs secured through adequate reimbursement.”
“Additionally, a companion diagnostic test must have a reasonable turnaround time, typically within one day, to avoid delays in providing test results to the oncologists making the treatment decision. IHC and Dako’s recently introduced IQFISH technology fulfill all of these criteria and also have the advantage that most pathology laboratories are performing these tests today, and therefore have the infrastructure, know-how, and skills necessary to perform these assays.”
Pedersen adds that since most new drugs are directed at a protein, and the protein represents the functional entity in the cell, it is important to look at the protein in the context of both cellular localization (membrane/nuclear/cytoplasmic) and being able to assess protein expression levels directly in the context of the tissue. “Again, IHC allows for this type of determination and performs very well,” Pedersen notes.
For the future, Pedersen agrees that NGS and quantitative polymerase chain reaction (PCR) may be used more in the clinical setting. “We are already seeing companion diagnostic collaborations that utilize quantitative PCR technology. At the moment, these technologies represent big potential, but also big challenges.”
“With the rapid pace at which the technology is moving, today’s NGS platform may differ from tomorrow’s NGS platform. It is uncertain what effect NGS will have on the quantitative PCR-type tests being developed today. However, it is crucial for pharmaceutical companies to select a technology platform for their companion diagnostic that won’t be outdated by the time the drug is launched.”
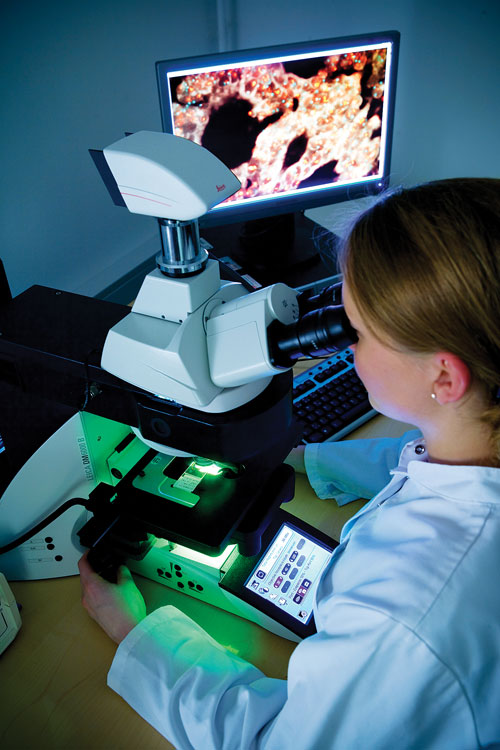
Some companion diagnostic assays make use of fluorescent in situ hybridization (FISH), as does Dako’s automated IQFISH system. The system depicted here shows an IQFISH slide beneath the lens of a fluorescent microscope, and fluor-escently labeled cells appear on a screen.
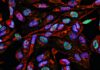
Epigenetic Markers Found for Head and Neck Cancers
Citing the potential for noninvasive tests capable of the early detection of head and neck cancer, researchers based at Queen Mary University in London announce that they have identified epigenetic changes in cancerous cells that are not seen in healthy cells. In particular, two genes (GLT8D1 and C6orf136) were found to be differentially expressed in head and neck squamous cell carcinomas (HNSCCs). Using methylation-specific quantitative PCR, the researchers confirmed that the promoters of GLT8D1 and C6orf136 were hypo- and hypermethylated, respectively, in HNSCC tissues.
The researchers published their results October 1 in the journal Cancer, in a paper entitled “Identification of FOXM1-induced epigenetic markers for head and neck squamous cell carcinomas.” In this paper, the researchers describe how they analyzed clinical specimens of malignant tissue from 93 cancer patients from Norway and the United Kingdom. These specimens were compared with either tissue donated by health individuals undergoing wisdom tooth extractions, or with noncancerous tissue from the same patients.
In their study, the researchers focused on FOXM1, an oncogene that has been implicated in all major forms of human cancer. They hoped to identify the earliest cancer-predictive epigenetic biomarkers accrued during aberrant cell proliferation induced by FOXM1. They identified a panel of FOXM1-induced differentially methylated genes, including C6orf136, MGAT1, NDUFA10, and PAFAH1B3, which were hypermethylated, and SPCS1, FLNA, CHPF, and GLT8D1, which were hypomethylated.
In addition, the researchers further investigated and validated the expression profiles of these candidate genes in head and neck tissue specimens. The researchers noted that they measured differential promoter methylation and gene expression in clinical specimens using commercially available bisulfite conversion kits and absolute quantitative PCR, respectively.
Of the four hypomethylated genes, three genes—FLNA (filamin A, alpha), CHPF (chondroitin polymerizing factor), and GLT8D1 (glycosyltransferase 8 domain containing 1)—were found to be consistently upregulated, although only GLT8D1 was statistically significant in both HNSCC cohorts. The researchers asserted that their promoter methylation analyses confirmed that the promoters of these three genes were indeed hypomethylated, a result that the researchers say suggests that promoter demethylation may be a mechanism responsible for gene expression hyperactivation: “Given that the expression of the three genes was aberrantly upregulated in both cohorts of HNSCC tissues, we speculate that they may play a role in oncogenesis.”
Of the four hypermethylated genes, only one gene, C6orf136, was found to be consistently downregulated in both HNSCC patient cohorts. Their methylation study, the researchers said, confirmed that the promoter of C6orf136 was hypermethylated in HNSCC, suggesting that downregulation of its gene expression may be a result of promoter hypermethylation.
Lead researcher Muy Teck-Teh, M.D., from the Institute of Dentistry at Queen Mary, said: “In this study we have identified genes which were either over- or underexpressed in head and neck cancer. The expression of these genes was inversely correlated with particular DNA methylation marks, suggesting the genes are epigenetically modified in these cancers.”
Dr. Tuck-Teh said that these epigenetic markers could be clinically exploited as biomarkers for early precancer screening of head and neck cancer, but he also noted that further work would be needed, as his group’s work is still in the discovery stage. At present, there is still no diagnostic test.
“The eventual aim would be to test asymptomatic patients and/or people with unknown mouth lesions,” added Dr. Tuck-Teh. “An advantage of epigenetic DNA markers is that it may be possible to measure them using noninvasive specimens. So it could enable the use of saliva, buccal scrapes, or blood serum for early cancer screening, diagnosis, and prognosis.”
Last year, in the International Journal of Cancer, researchers from Queen Mary University published a paper in which they described a genetic test that they had developed for the early detection of oral cancer. That test, called the quantitative Malignancy Index Diagnostic System, quantifies the expression levels of 14 genes associated with the FOXM1 cancer gene, and converts the measurements into a diagnostic score that indicates the risk of a lesion becoming cancerous.
This work, said the researchers, showed that aberrant upregulation of FOXM1 “brainwashed” normal cells by reprogramming the methylome, changing its landscape toward those found in cancer.
Epigenetic markers for head and neck squamous cell carcinomas have been identified that could be clinically exploited as biomarkers for early precancer screening. [Eraxion/iStock]