July 1, 2009 (Vol. 29, No. 13)
Krista Witte
ForteBio’s Dip and Read Method Simplifies Label-Free Assays
Molecular interactions play a myriad of important roles in drug discovery and development. From determining mechanism of action to identifying unwanted biological reactions (e.g., monitoring for an immune response to an administered drug), the study of binding events is a critical part of many phases of therapeutic research.
Successful analysis of molecular binding kinetics requires the ability to bring two molecules in close proximity under conditions that can overcome diffusion limitations. Much of the commercially available biosensor instrumentation has relied on microfluidics to flow a sample containing the analyte of interest over a surface with the binding partner immobilized. While providing efficient flow, the use of microfluidics limits the sample types that can be used due to the issues of clogging and fouling. In addition, organics and proteins can bind to the large surface area of the microfluidic system contaminating subsequent samples.
To overcome these disadvantages, a novel dip-and-read instrument platform has been developed. ForteBio’s Octet system uses a fiber optic based biosensor surface that is dipped into the sample of interest. By moving the biosensor surface to the sample, as opposed to moving the sample to the surface, cross contamination from previous samples can be minimized. Samples, buffers, and reagents all remain in the standard 96-well or 384-well microplate throughout the assay while up to 16 biosensors are simultaneously dipped into each reagent in turn.
To overcome diffusion limitations, the microplate containing the samples is moved in an orbital motion relative to the biosensor. Molecular binding kinetics determined using this orbital flow in the Octet platform agree well with those determined using more complex microfluidics based instrumentation.
The Octet instruments utilize biolayer interferometry (BLI) to monitor the increase or decrease in optical thickness of molecules on the fiber optic biosensor surface (Figure 1).
BLI is an optical technique that analyzes the interference pattern of white light reflected from two surfaces: a layer of immobilized protein on the biosensor tip and an internal reference layer (Figure 1). Any change in the number of molecules bound to the biosensor tip causes a shift in the interference pattern that can be measured in real time. By monitoring the change in molecular thickness, the binding and dissociation of molecules from solution to a surface-immobilized capture molecule can be observed and rates of association and dissociation can be calculated.
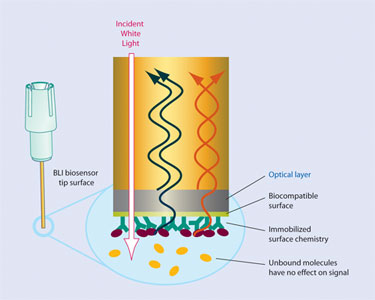
Figure 1. Biolayer interferometry
Macromolecules and Particles
Proper assay design is critical to the success of any interaction analysis. The molecule to be immobilized onto the surface should be of high purity and activity. The protein can either be immobilized using a direct coupling method or biotinylated and immobilized onto a Streptavidin-containing surface. Direct coupling and biotinylation using NHS reagents both react with the same amine residues on the protein to be immobilized.
Since biotinylation can be controlled by optimizing the ratio of biotin-NHS to protein in solution, immobilizing through biotinylation is preferable when dealing with a delicate protein. In addition, using a biotinylation reagent with a long, flexible linker can also help the protein be more accessible for subsequent binding by the analyte.
Kinetic analysis of large molecules such as proteins, DNA, or even larger particles such as phage or microspheres can be done using the Octet. The lack of fluidics, along with the fact that the BLI technology is minimally influenced by molecules in solution, allows for a wide range of sample types including lysates, cell culture supernatants, purified, and phage preparations to be used.
Small Molecules
Kinetic analysis of small drug-like molecules binding to protein targets is a crucial aspect of drug discovery. Small molecules present several unique challenges for interaction analysis. Most obvious is their size, which is typically less than 500 daltons, resulting in small binding signals and low signal to noise. They also exhibit fast association and dissociation rates with proteins, the accurate measurement of which can be challenging. In addition, small molecules are generally less soluble in standard aqueous buffers than proteins and require the addition of organic co-solvents such as DMSO.
To combat the low signal to noise seen in small molecule experiments, two pathways have been pursued simultaneously. New super streptavidin (SSA) coated biosensors with increased binding capacity for biotinylated capture molecules increase signal three- to eightfold depending on the molecular system. In addition, higher sensitivity spectrometers allow the newest Octet instruments (Octet RED and Octet RED384) to have an eight- to tenfold reduction in noise. Together, the two approaches have enabled the analysis of binding kinetics of small molecules as small as 157 daltons.
The use of cosolvents at levels that are compatible with most proteins (1–5% DMSO) are easily tolerated in BLI-based analysis, but should be kept consistent throughout the assay. In Figure 2, kinetic analyses of three different known inhibitors for carbonic anhydrase are shown. Biotinylated carbonic anhydrase was immobilized onto the SSA biosensor surface. The analysis for each inhibitor was performed in duplicate at five different concentrations in 5% DMSO. Kinetic constants determined for these inhibitors agree well with literature values.
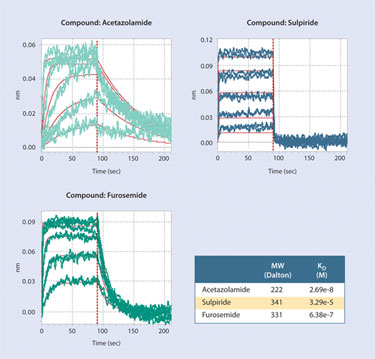
Figure 2. Analysis of known inhibitors binding to carbonic anhydrase
Immunogenic Response to a Drug
Interaction analysis can also be used to monitor unwanted binding events such as the detection of undesirable immune responses to a therapeutic drug. The immune system may respond to drug administration in patients by producing antidrug antibodies (ADA). These ADAs could alter the pharmacodynamics and pharmacokinetics of the drug. ADAs can be produced in a wide range of concentrations and with a wide range of affinities. To accurately detect these antibodies, the assay must have good sensitivity, withstand a wide range of free drug concentrations, and have minimal matrix effects.
The Octet system can be used with the available Dip and Read Immunogenicity Kit to detect ADAs in both human and animal samples. The assay is run without plate-washing steps allowing detection of low affinity antibodies that could be missed in ELISA type assays. Figure 3 shows data that demonstrates the ability of this assay to tolerate 500-fold excess of drug and still have sensitivity to 20 ng/mL of ADA.
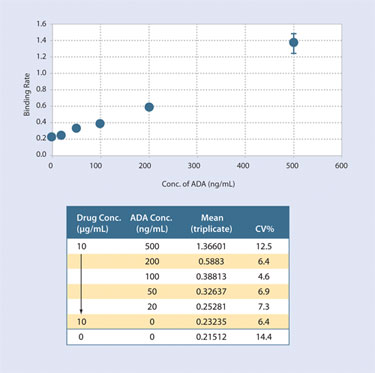
Figure 3. Resulting data from the enzyme–linked bridging assay protocol using the immunogenicity kit on the Octet RED
Krista Witte, Ph.D. ([email protected]), is senior director of chemistry and consumables R&D at ForteBio. Web: www.fortebio.com.



