April 15, 2012 (Vol. 32, No. 8)
Vicki Glaser Writer GEN
The initial resistance that the high-content/high-throughput community had to face has been overcome, and now most in the field acknowledge that flow cytometry is enormously high throughput and very high content.
One of those converts is J. Paul Robinson, Ph.D., SVM professor of cytomics and professor of biomedical engineering at Purdue University. At CHI’s recent “High-Content Flow Cytometry” meeting, Dr. Robinson talked about the ability to automate flow cytometry-based high-content screens and data processing to perform multiplexed high-content analysis in real-time, running multiple assays and samples in parallel, and to miniaturize the technology to conserve resources and reduce costs.
Dr. Robinson also discussed how data-extraction issues have historically been a stumbling block in the use of flow cytometry in drug discovery. Dr. Robinson described the main challenge in streamlining multiparametric data processing as eliminating operator input and avoiding manual gating. Automation provides a solution to managing the huge datasets produced with multiparametric high-content analysis.
To overcome the inefficiencies of traditional approaches to data analysis, Dr. Robinson’s group developed a technology based on a sequential gating process. The method involves combining all of the data files acquired into one file for analysis. This not only accelerates the process but also facilitates data comparison.
Using this approach, a biological sample can be analyzed in parts—exploiting the ability to separate individual cells for analysis using flow cytometry—with the data then combined to piece together the complete picture. Automation of the entire process—from sample preparation, to running complex assays, to data analysis—makes it possible to perform functional phenotypic profiling of cell populations efficiently.
The most important factors, he explained, are to embed multiple computational algorithms and analytical tools into the data analysis software and to extract the feature space correctly. He described the graphical interface the group developed—a logic map designed to analyze the data from functional live-cell assays that incorporate fluorescent markers.
In addition, Dr. Robinson presented an example of multicolor flow analysis to demonstrate the ability to perform cytomics studies for pathway analysis. Overall the use of flow cytometry to analyze individual cell populations and evaluate the effects of drugs on cell function allows you “to ask the what-if questions” and to begin to integrate pathways, he said.
Peter Krutzik, Ph.D., a senior scientist in Gary Nolan’s group at the Baxter Lab in genetics, pharmacology, microbiology, and immunology at Stanford University, described the use of phosphor flow cytometry to profile perturbations in signaling networks when peripheral blood cells are exposed to drug compounds.
This method allows for multiplexed analysis of cell types and signaling pathways, and has applications in drug target identification and throughout the drug development workflow, including in the analysis of patient samples during clinical testing.
The increasing use of automation and miniaturization is lowering the cost of flow cytometry-based high-content analysis in primary and secondary drug screening, making it possible to run larger screening campaigns.
Describing the benefits of using flow cytometry to study disease specificity as it relates to cell types and signaling pathways, Dr. Kritzik reported that it is possible to measure at least 10 parameters in 5,000–10,000 single cells per second. This capability is particularly important when studying heterogeneous cell populations and a variety of different phosphoproteins.
Dr. Kritzik and colleagues are using phosphor-flow cytometry to study three phosphoproteins—all linked to disease and all potential drug targets—in subsets of peripheral blood cells. Taking advantage of the capacity for multiplexing, they can perform 14 assays per well, generating reproducible data with low intra- and inter-run variance.
The screens have demonstrated cell-type specific druggability; for example, a particular drug might inhibit kinase activity in T cells but not in monocytes. The researchers were able to correlate cell populations with pathway activity. Grouping the data according to a specific phosphoprotein yields information on selectivity patterns.
Furthermore, Dr. Kritzik presented data from an automated secondary dose-response screen in which they were able to generate more than 20,000 data points per day and to create IC50 fingerprints for various Jak kinase inhibitors that are currently in development in the pharmaceutical industry.
For a review of a paper published about an approach to high-content methods that enables pathway analysis from primary cell types through atomic mass cytometry, click here.
Optimizing Data Analysis
In his presentation, Dan Ehrlich, Ph.D., research professor of biomedical engineering at Boston University, emphasized the problems associated with segmentation in data analysis and the “noise” it can introduce due to boundary drawing errors.
Various algorithms can be used to perform cell segmentation in the analysis of data from flow cytometry-based imaging studies. In segmentation, individual pixels are classified as belonging either to a particular region of the cell or to the background, and misclassification errors can affect a variety of measurements and calculations.
Dr. Ehrlich’s team has developed a method for one-dimensional high-content screening in a flow system using a parallel microfluidic cytometer (PMC), which incorporates a high-speed scanning photomultiplier-based detector, for low-pixel-count, one-dimensional imaging. He described published work using the system to generate six-pixel 1-D images in studies of protein localization in a yeast model for human protein misfolding diseases and in nuclear translocation assays in CHO cells expressed in an NFκB-EGFP reporter.
His group is developing a next-generation, simpler 1-D flow system and experimenting with modifications such as increased scanner resolution, various ways of implementing flow-based high-content analysis, and the potential for clinical applications of PMC in a disposable chip format.

Scientists at Boston University have developed a method for one-dimensional, high-content screening in a flow system using a parallel microfluidic cytometer.
Enabling High Throughput
Larry Sklar, Ph.D., director, University of New Mexico Center for Molecular Discovery, pursues research on the application of flow cytometry for drug discovery and explores the molecular interactions at cell surfaces that lead to intracellular signaling. His laboratory is a member of the Molecular Libraries Program of the NIH Roadmap Initiative.
In his presentation, Dr. Sklar described his group’s use of a 384-well plate-based HyperCyt® high-content flow cytometry screening system from IntelliCyt. In development is a 1,536-well system capable of generating 250,000–500,000 data points/day. Their research focuses on the regulation of integrin, an adhesion molecular present on the surface of white blood cells.
Dr. Sklar and his team are conducting high-throughput, high-content screens to evaluate the ability of small molecules and repurposed drugs to activate signaling pathways that lead to changes in the conformation and affinity of integrins.
Dr. Sklar said that one of the most powerful aspects of flow cytometry for these types of studies was the ability to use a laser beam to generate a signal from a fluorescently labeled ligand that interacts with integrins on the leukocyte surface and detect in real-time associated changes in integrin affinity and cell activation that enable the leukocyte to interact with an endothelial cell. This technique enables studies of association/dissociation kinetics and the ability to experiment with interventions intended to modulate the dissociation rate.
The researchers have also developed fluorescence resonance energy transfer (FRET)-based assays to detect integrin activation and extension, using the data to correlate changes with agonist stimulation of various GPCRs, and apply the information gained on differential integrin regulation to pathway analysis. This knowledge could then feed back into the design of drug screens to identify small molecules that activate targeted pathways.
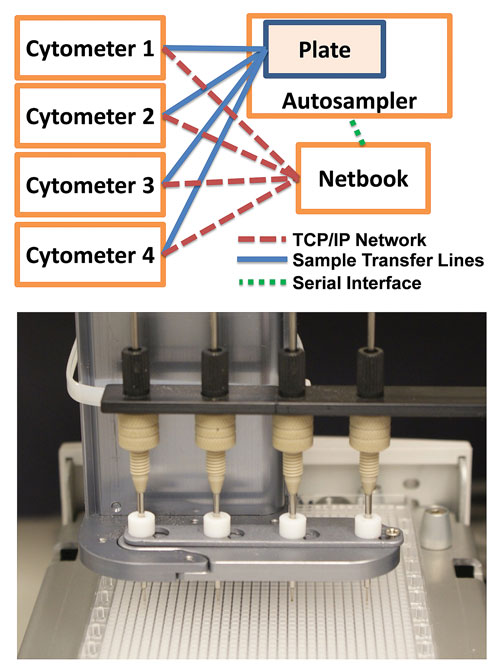
1,536-well plate-based high-content flow cytometry screening platform developed by IntelliCyt and researchers at the University of New Mexico. Top: The Cluster Cytometer HyperCyt platform illustrating sample transfer lines linking sample uptake ports from each of four flow cytometers to the four sampling probes positioned above the 1,536 well plate (solid lines), and serial (small dashed lines) and Ethernet network (large dashed lines) connections by which a netbook PC controls the autosampler and cytometers, respectively. Bottom: Four sampling probes during processing of a 1,536-well plate.
Tools for High-Content Cell-Based Assays
Exhibitors at CHI’s “High Content Analysis” conference featured a range of technologies and products to facilitate and improve live-cell functional assays capable of high-resolution cellular and sub-cellular visualization.
CYTOO plates from CYTOO Cell Architects are high-resolution glass-bottom microplates with thousands of adhesive micropatterns arrayed on the cytophobic surface in each well. Cells attach to the concave surface, stretching across the nonadhesive areas, forming micropatterns. The geometric distribution of the adhesive contacts controls the polarization of the cell, normalizing cell position, shape, polarity, and the organization of internal cell components.
Thermo Fisher Scientific highlighted its ArrayScan VTI HCS Reader with the CrEST X-Light Confocal Module that contains a high-speed Nipkow spinning disk and offers four-color illumination, a user-definable z-range, and software-controlled switching between 40 µm and 70 µm pinholes.
Expanding on its long-standing partnership with Genedata, Thermo Scientific and Genedata recently introduced the Genedata Screener® Cellomics Edition data mining and visualization tool for high-content screening. Genedata’s Cell Population Extension for the Genedata Screener enables interactive processing of single-cell data from high-content screens. The software calculates the results per well from individual cell populations based on user-defined criteria, and aggregates cell values to generate an overall result for the population.
Curiox Biosystems presented its DropArray™ technology, which the company describes as a replacement for traditional microarrays. The platform comprises DropArray plates—“wall-less” 96-well or 384-well plates with a hydrophilic “well” surrounded by a hydrophobic area—and the DropArray Accelerator HT washing station for high-throughput processing of cell-based assays, including the use of suspension or semi-adherent cells. Advantages include the absence of edge effects, gentle washing, and small sample volumes (2–4 µL).
Amnis, recently acquired by EMD Millipore, offers the ImageStreamX®, a high-resolution microscope for imaging suspended cells capable of producing brightfield, darkfield, and fluorescence images, with quantitative image-analysis standards. New to the company’s product line is the FlowSight, a two laser/12 channel imaging system that can be configured with up to four excitation lasers, a 96-well plate AutoSampler, and optional quantitative image processing.
Fluofarma featured its CytoSurfer® image data-analysis software for high-content screening of 96- or 384-well plates. It provides a heatmap view, dose-response functions, and a batch-processing tool. The software uses interactive multidimensional scatterplots, histograms, and well images to visualize data, allowing users to go back and forth between the data and the original cell images. Users can select and independently analyze individual cell populations.
GE Healthcare Life Sciences featured the IN Cell Analyzer 6000 laser-based confocal imaging platform for high-content analysis, as well as the DeltaVision OMX BlaZe super-resolution system from Applied Precision (a GE Healthcare company). The OMX BlaZe incorporates a fast structured illumination module, three sCMOS cameras, and five lasers, with an optional fourth camera and additional lasers for 3D-SIM and Widefield imaging.
Vala Sciences developed the kinetic image cytometer (KIC) as an add-on to its IC200 high-content screening system. The KIC puts an electrical probe in each well of a microtiter plate to enable combined high content analysis and electrophysiology studies with measurements of individual cell kinetics. The ability to study cellular calcium transport and to perform cell-by-cell analysis of calcium transient dynamics in an automated manner makes it possible to do high-throughput screening of calcium channel activity associated with drug-related cardiac arrhythmia liabilities.
Wako Laboratory Automation launched the Cell Voyager CV7000 to the U.S. market for live-cell, high-content imaging. The platform offers a S-CMOS camera cooled to -30°C for noise reduction, a stage incubator, and spinning disk technology that eliminates photobleaching. The system performs three-color simultaneous imaging and captures four images per well on a 384-well plate in four minutes. It is also capable of performing nonconfocal imaging, including phase contrast and epifluorescence.
Black Mesa Imaging develops customized, platform-agnostic software to meet a user’s specific needs for computational analysis. The company’s Prometheus image-analysis software performs automated processing of digital image files, detects and graphically displays objects and features, and records measurements.
Intellicyt featured its HTFC Screening System, a high throughput flow cytometry system with continuous “stream-bubble delineated” sample loading that can load a 96-well plate into the system in 3 minutes and a 384-well plate in 12 minutes. This benchtop analyzer collects four-color fluorescence and two light scatter measurements on individual cells simultaneously and is capable of screening up to 40 wells per minute. The system includes ForeCyte software and IntelliCyt Data Manager (iDM).
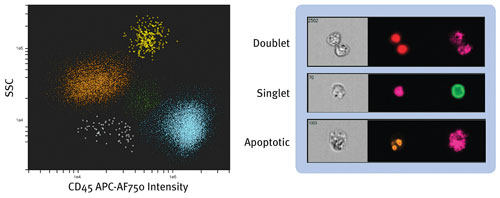
Amnis reports that its FlowSight imaging flow cytometry combines the best of flow cytometry and microscopy: Dot plot of human peripheral blood mononuclear cells partitioned into five distinct populations. Multicolor images of every cell permit visual verification and quantitative image analysis.



