January 15, 2011 (Vol. 31, No. 2)
Chris Torrance, Ph.D.
Genetically Defined X-Man Disease Models Find Niche in Personalized Medicine Applications
The output of high-throughput genome sequencing technologies has been accelerating at an exponential rate and, consequently, the full-scope of human genetic variation is now within our reach. The technical bottleneck is now moving to “what does it all mean?” This crucial question is the challenge of functional genomics, an increasingly important field that will need both advances in human genetic manipulation techniques and large consortium-based research efforts to decipher how specific genetic variations predispose or accelerate human pathogenic disorders.
In pursuit of these aims, Horizon Discovery is commercializing (to biotech, pharma, and diagnostic companies) or providing in open access (to academic research organizations) a gene-editing platform called Genesis.
This technique is built on the Nobel Prize winning discovery that homologous recombination (HR) can be harnessed to perform precise genome alterations in mice. Genesis takes this technique many steps further to permit efficient gene editing in any pre-established and differentiated human cell line, which in contrast to mouse ES cells, have low rates of homologous recombination. This is achieved using recombinant adeno-associated (rAAV) vectors which, due to their ssDNA genome, activate HR without resorting to causing dsDNA breaks in the genome, typical of zinc-finger nucleases (ZFNs) and meganucleases.
Moreover, because dsDNA breaks are preferentially repaired by error-prone non-homologous end-joining pathways, ZFNs are more appropriate for performing loss-of-function gene knockouts. In contrast, rAAV can readily perform both knockout and gain-of-function knockins, enabling the modeling of subtle SNPs or activating point-mutations that are more commonly the targets of drug discovery programs.
To date, Horizon has used Genesis primarily to generate a large and expanding panel of genetically defined X-Man (mutant and normal) disease models. These are matched pairs of cell lines, wherein one harbors a cancer-associated mutation in an endogenous gene, just as it occurs in real patients, and the other is an otherwise genetically identical cell line except that is carries a normal version of that gene.
These isogenic disease models provide a definitive means to understand disease biology and develop new targeted therapies to specific patient populations.
One of the most exciting applications of X-Man cell lines is to predict, prior to entering clinical trials, which patients will benefit most from a new targeted therapy. The benefits of tailoring the right drug to the right patients in clinical trials will inevitably lead to faster approvals, more ethical patient accrual, and less chance of drug effects being missed in larger unselected patient cohorts. Such information is essential in order for healthcare agencies to ensure that only patients with a rational and diagnosable predictive biomarker are a given a specific medication.
Colon cancer patients with K-Ras mutations were recently assessed in retrospective and prospective studies to be nonresponsive to EGFR-therapies Erbitux and Vectibix. As a result, testing of K-Ras mutational status is now mandatory prior to prescription of these agents. It should be noted, however, that there are several different mutational variants of K-Ras and currently a patient carrying any variant K-Ras is excluded from anti-EGFR therapy in both the EU and the U.S.
In an attempt to provide greater resolution to the influence of specific K-Ras mutational biomarkers, and in response to a small number of clinical anomalies to this resistance profile, Horizon’s co-founder, Alberto Bardelli, set out to test two X-Man cell lines harboring the most predominant K-Ras mutations (G13D or G12V) for their response to Erbitux.
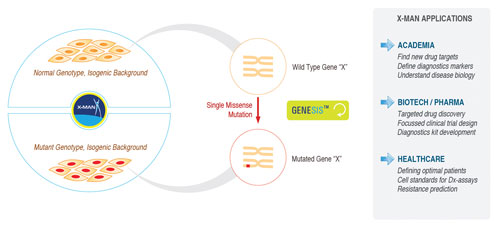
Figure 1. Genetically defined X-Man cell lines and their applications
Methods
K-Ras wild-type and Erbitux-sensitive SW48 colon cancer cells were selected for engineering using the Genesis platform, wherein two different variants of K-Ras mutation (G13D and G12V) were spliced into the endogenous K-Ras locus. These two isogenic cell lines and WT parental cells were then tested for their sensitivity to Erbitux in vitro and in vivo.
Results
In both in vitro proliferation assays and in xenografted tumors, G12V containing SW48 cells were found to be completely unresponsive to Erbitux. However, G13D containing SW48 cells, along with the parental WT SW48 cells, were highly responsive to Erbitux. This experimental data was unambiguous, showing clearly that G13D mutations do not impart resistance to Erbitux therapy in these patient-relevant disease models. Bardelli and colleagues were thus prompted to subsequently perform a new and larger retrospective sequence analysis on colon cancer patient tumor samples for their K-Ras mutational status and compare this with their known clinical response data to Erbitux.
This patient study demonstrated a clear concordance with the X-Man disease model data and confirmed that G13D patients are benefitting from anti-EGFR therapy. Prospective clinical trials will need to be performed to confirm this suspicion and, if conclusive, may lead to a further refinement in the rules for prescribing EGFR-targeted therapies in colon cancer.
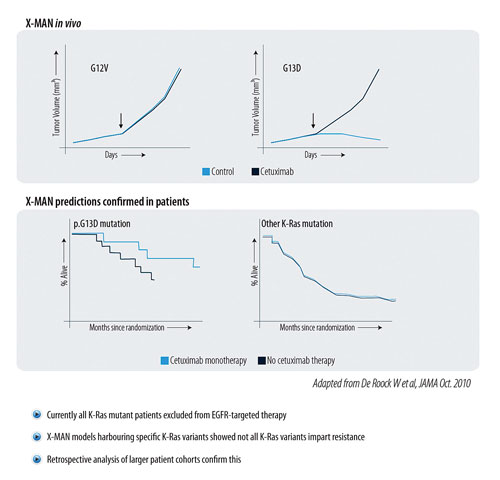
Figure 2. Predicting K-Ras resistance to EGFR-targeted agents in colon cancer [De Rook W., et al., JAMA Oct. 2010]
Conclusions
X-Man cell lines are a powerful tool to predict patient responses to targeted therapies and they provide a definitive understanding of the genetic factors that mediate their sensitivity or resistance to such agents. Further in vitro studies are now ongoing using a suite of SW48 based X-Man cell lines covering several other variations of mutant K-Ras and other genotypes for their effects on EGFR-targeted agents.
Other targeted agents are also being tested in this system so as to potentially inform the prospective integration of clinical biomarkers into new clinical trials. Expanding on this ambition, Horizon and several translational and clinical collaborators have been awarded a large consortium grant to define and integrate new genetic biomarkers into X-Man models and build algorithm for prospective clinical trials.
Genetically defined X-Man human cell lines are important tools to predict which patients will benefit from new targeted or personalized medicines. Building upon the K-Ras/Erbitux data, examples of personalized medicine will become increasingly manifold as such models are extended upon and increasingly disseminated within the academic and industrial research community.
Figure 3. X-Man genetically defined and patient-relevant human cell lines
Chris Torrance, Ph.D. ([email protected]), is CSO at Horizon Discovery.







