August 1, 2007 (Vol. 27, No. 14)
Gunter Jagschies Ph.D. Strategic Customer Relations Leader GE Healthcare Life Sciences
Ann O’Hara
Real Issues Are Flexibility in Manufacturing and Uncertainty about Future Demands
Biopharmaceutical manufacturing involves a balancing act between the capacity of cell culture and downstream purification. Balancing the two has always been difficult because construction decisions are taken 3–5 years before production is started. Hence, by definition, plants coming online today are designed on the technology and yield assumptions from 3–5 years ago.
In fact, over the past 20 years, it would appear that the productivity improvements from increasing titres in upstream have created a challenge in downstream purification. But when one looks into the math it becomes obvious that downstream technology was significantly more capable than upstream technology until just a few years ago, and recent developments prove it is just as capable today, despite the popular downstream bottleneck misconception. The real issues are flexibility in manufacturing and uncertainty about future demands.
As an example, at the turn of the century, if you were to plan to produce 500 kg per year of a particular protein, your manufacturing suite would contain 9 or 10 of the largest commercially available bioreactors but only need three downstream process trains. This is based on the following formula, one 10,000-L bioreactor could produce roughly 60 kg per year (15 batches/year x 10,000-L bioreactor x 500 mg/L titre x 80% yield/1,000,000 mg/kg). And, on the downstream side, one process train could purify roughly 240 kg per year (65 runs/year x 300-L column x 15 g/L capacity x 80% yield/1,000 g/kg).
To check this example, one may study the Enbrel™ (Amgen) case. Following first approval in November 1998, demand for this new drug soon exceeded expectations, and production capacity started lagging. Even though the estimated scale was still below 1,000 kg per annum, several production sites, each with multiple large-scale bioreactors each, had to begin production of this protein.
Why then is there a public debate about downstream bottlenecks? Because of the following: plant equipment is fixed, bioreactors are generating more product at increasing product titre, and the downstream capacity, designed previously for low upstream productivity, needs to be increased to match the new upstream productivity.
Upstream improvements in the last 20 years have led to a dramatic development from our example of 500 mg/L product titre it has now increased to 2–3 g/L in production, and we see 5 g/L in clinical manufacturing projects.
The same bioreactor in an existing facility can now potentially make fivefold more per batch with little change to the installations. Separation technology can handle these quantities, with more process trains or at larger column volume or membrane area. But it wouldn’t be necessary to increase fivefold, because during the same time period the productivity of chromatography resins used in capture steps has increased at the same rate through both higher capacity and processing speed.
So downstream technology can handle the purification scale, but the industry underestimated the capacity required. While one 20,000-L bioreactor will soon be capable of producing nearly 1,000 kg per year, one downstream line is already capable of doing this today.
Run at its best capability (modern resin, largest column size, highest column height), one downstream line can process product from six bioreactors of up to 15,000 L, each operating at the still-to-be realized product titre of 5 g/L.
Retrofitting Plants
The main reasons that bottlenecks are a topic for discussion are the time and costs involved to retrofit and revalidate existing plants. The portion of floor space in a plant occupied by upstream and downstream installations has shifted due to the much higher liquid consumption for buffer preparation and the need for storage capacity that supports a modern, large-scale downstream process.
As soon as such a modern process is to be accommodated into an existing facility of an older design, it requires retrofitting. Retrofitting a plant is a matter of priorities and payback. If a process is showing dramatic upstream improvements, it will likely be worth the expense to install more downstream capacity, and at the same time implement process improvements (changes!) even downstream.
Floor space required for the increased buffer volumes and their storage can be mitigated by using buffer concentrates and in-line buffer mixing so that tank size is reduced. Additionally, two-step purification process designs seem more feasible now than a few years ago. Resins have been introduced that show novel selectivities and seem able to address what previously required three chromatography steps. A two-step process occupies less floor space and consumes less buffer and thus addresses one of the key issues in existing facilities very well.
There are still a number of practical challenges to match a 2–3 week batch time on the upstream side with a 2–5 day batch time on the downstream side. Frequently up to six fermentors are operated in parallel, loading one batch every second day onto one and the same downstream line. A regularly used approach divides each downstream batch into a number of cycles that are processed sequentially in the capture step.
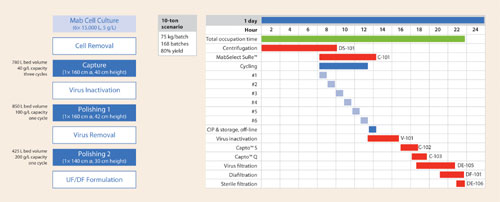
Figure 1 presents a process scheme with column dimensions for a hypothetical 10-ton scenario and a GANTT chart assuming 24-hour operation. Even with a less intensive two-shift operation, the total time for a downstream batch can be as short as two days, down from at least four to five days with first-generation resins. With this approach, one can better align upstream and downstream processing.
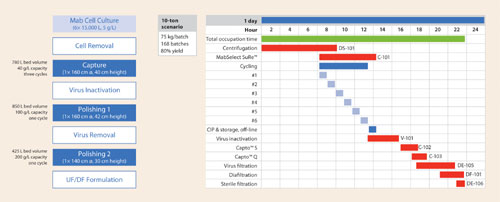
Figure 1A and 1B
Demand for Drug Volume
One thing the industry has learned over the last 20 years is the certainty of uncertainty. Concurrent with trends in increasing upstream productivity, we have seen the average demand for drug volume per application decrease. Today, few monoclonal antibodies require an annual production of beyond 1,000 kg. Examples of mAbs reaching the ton scale are Rituxan™ and Avastin™, marketed by Genentech and Roche respectively, but in most cases the scale of manufacturing is between 50 and 500 kg. And there is little indication that this will change in the future.
Few medical indications will be large enough to create ton-scale demands, and such indications are under heavy competition, decreasing the demand for each branded medicine. Similarly drugs are becoming more potent, further reducing the need for kilograms. So why build such large process equipment if a 20,000-L bioreactor could produce to demand with just a few batches per year and be significantly underutilized with that challenge?
While there is a need for a handful of large-scale manufacturing plants, new facilities must be designed for flexibility that will allow for varying demands per medicine, fast change-over between production campaigns, and production of multiple products both in series and in parallel.
Disposable equipment that includes separation devices and plug-and-play hardware will be elements of such a design. The largest bioreactor in such a facility may be small relative to current installations. Batch time would be developed toward “shorter than today.” Together these elements would allow for the unexpected to happen and be successfully handled.
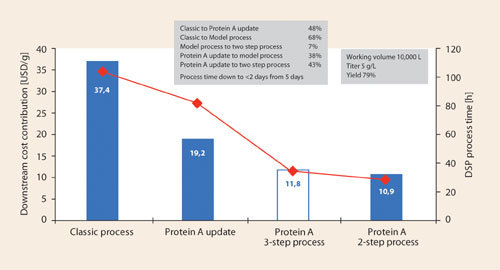
Figure 2
Economics
A quick look at the P&L statements from major producers of protein-based medicines seems to indicate that manufacturing costs for such drugs are not as high as one might expect. Both Genentech and Amgen are mainly marketing protein drugs and report a manufacturing cost of around 15% relative to their revenue (2006 data).
If we reverse this calculation and subtract known overheads, it appears that the production costs for mAbs fall into a range between $100 and $200 per gram as long as good facility utilization is assumed. The overall downstream part may account for 25 to 30% of total costs. So why is there talk of strong cost increases for the downstream process (in absolute dollar value)? With higher titres from the bioreactor, each downstream step needs to be larger and is producing more Kg of antibodies, and so it appears to be more expensive in total. But the costs per gram have been going down.
Figure 2 shows model calculations for monoclonal antibody processes using different generations of purification resins. The classic process uses resins of the Sepharose™ Fast Flow generation. Updated processes use resins of the MabSelect™ and Capto™ generation. Cost reductions of more than 60% can be achieved by introducing modern resins. This is a joint effect of improved binding capacity, longer life time, and much reduced batch time.
Apart from costs, revenue determines financial performance of any business. Enbrel is one case of a biopharmaceutical where revenue generation was compromised by lack of production capacity.
The lesson from this case is twofold: for the manager to avoid production bottlenecks whenever there are good opportunities for all product to be sold, and for the process developer to focus on improving process yield as one form of increasing output from the facility.
Every percent of increased yield can generate many millions of dollars of additional revenue. Obviously, similar considerations apply to the demands on the plant engineer to ensure flexibility and increased facility output through use of modern design concepts adaptable to a rapidly changing environment.
In summary, one may conclude that the true bottleneck is not due to the technology we have today to produce biopharmaceuticals. The real issues are related to the inflexibility of available facilities and process designs, regulatory hurdles to change and improvement, and the difficulty in predicting future demands.
There is one prediction, however, that can be made: manufacturing of future generations of biopharmaceuticals will be both possible and cost-efficient. Improvements in downstream processing technology will further support this. Likely improvements will include higher capacity capture resins, separation devices and formats for highly flexible facilities, and semicontinuous processing methods removing wasteful activities from the process flow.
Günter Jagschies, Ph.D. ([email protected]), is senior director of biopharma technology, R&D, and Ann O’Hara ([email protected]) is general manager of bioprocess at GE Healthcare Lifesciences. Web: www.gelifesciences.com/bioprocess. Capto, MabSelect, and Sepharose are trademarks of GE Healthcare companies. All third-party trademarks are the property of their respective owners.