December 1, 2012 (Vol. 32, No. 21)
Vicki Glaser Writer GEN
Pushing the Limits of Imaging Speed and Resolution
In any field of science, new discoveries often depend on finding answers to fundamental questions. In drug discovery, that often means uncovering the differences between healthy and diseased cells and tissues and understanding the biological basis of cellular function and dysfunction at the molecular level.
Microscopes and imaging technology have evolved in synchrony with advances in computers and software applications, detectors, light sources, and labeling and detection systems. Microscopes have an increasingly important role in research aimed at illuminating what exactly happens, for example, when a virus infects a host cell, a ligand binds to a membrane receptor, or a tumor cell metastasizes.
The abundance of published articles featuring advanced imaging techniques, and grant awards to support the establishment and expansion of imaging centers are evidence of the critical role of microscope technology in biomedical research.
For example, The HIV Center at the University of Utah recently received a $21.8 million grant from the NIH to develop methods to image and study the structural biology of the AIDS-causing virus and to understand how it infects host cells, replicates, and spreads.
The Molecular Imaging Center at Washington University School of Medicine in St. Louis received a $7.1 million grant from the NCI. And the Dana-Farber Cancer Institute received a $10 million grant from the Massachusetts Life Sciences Center to expand its cancer imaging research program and to help establish the Molecular Cancer Imaging Facility, a research initiative aimed at developing new molecular imaging probes.
These sizable grants also emphasize the high cost of imaging technology, and one overarching goal expressed by developers of microscope systems is to bring down the cost of ownership and expand accessibility of the technology.
While the most advanced technology is available to the top biologists in the world, “we need to make it more financially and technically accessible to all biologists,” says Paul Goodwin, science director at Applied Precision, a GE Healthcare company.
In surveying the microscopy industry, several key trends emerge, most prominently an emphasis on increased resolution and the emergence of super-resolution technology and dynamic imaging in live cells and organisms.
The super-resolution methods that have emerged represent an alphabet soup of sophisticated techniques including structured illumination microscopy (SIM), stochastical optical reconstruction microscopy (STORM), spatially modulated illumination (SMI), ground state depletion (GSD), and stimulated emission depletion (STED).
Live-cell/organism imaging requires faster image acquisition to capture biological and physiological processes as they are happening, such as protein-protein interactions, cell migration, morphologic changes related to cell division or apoptosis, and ion-channel activity. Growing interest in live-cell imaging has spurred developments in related fields such as label-free imaging to enable less intrusive and disruptive methods that do not require exposure to large amounts of light likely to affect the biology and compromise the viability of cells.
Additional trends focus on enhanced quantitation, co-localization, and correlative microscopy, and the integration of different microscopy techniques in unified multifunctional systems. Advances in deep-tissue imaging and 3D microscopy reflect a trend in cell biology away from studying cells grown in monolayers to examining multicellular and tissue constructs to explore biology in a more natural, dynamic environment.
The challenges lie mainly in the ability of tissues to absorb and scatter light, and other factors that contribute to poor signal-to-noise ratios when imaging through tissues and aberrations that can compromise resolution.
Resolution Solution
Goodwin describes a “renaissance in microscopy,” with multiple new technologies emerging across a relatively short time span to meet a variety of technical and application-oriented needs in the biopharma R&D arena.
When you get down to basics, “the glass lens of a microscope has not changed substantially in 100 years,” he explains. What has largely driven advances in microscopy systems has been the combination of sophisticated computational and sensor technology. This is evident across several of the key trends highlighted in this article, including super-resolution imaging technology, label-free imaging, and advances in correlative, quantitative, and 3D microscopy.
Advances in super-resolution imaging are enabling spatial and temporal resolution in the same sample and live-cell imaging, notes Goodwin, highlighting the company’s DeltaVision OMX® super-resolution system with the BLAZE™ 3D SIM module capable of imaging a 1 micron slice in <1 sec and widefield imaging at >400 fps.
He draws attention to a recently published paper by Strauss et al. (PLoS Biol), which described the use of 3D-SIM super-resolution microscopy to perform time-lapse imaging studies of cell division in bacteria and visualize protein dynamics and changes in protein distribution that contribute to cytokinesis.
Selecting a super-resolution technique can mean a trade-off between higher resolution versus dynamic imaging, explains Chris Vega, Ph.D., marketing manager, life science research at Leica Microsystems. With STED, for example, “you can image in a purely optical way” and typically achieve resolution in the 40−50 nm range, whereas with the GSD method, which involves the localization of individual fluorophores, higher resolution of about 20 nm is feasible using existing technology, but GSD performs better with fixed specimens than live samples.
Leica’s latest addition to its super-resolution systems is the third-generation TCS SP8 STED instrument, which the company introduced at the Society for Neuroscience meeting in October. It includes a gated STED option that combines Leica’s HYD™ hybrid detection technology and white light laser to increase resolution and contrast.
Is the microscopy industry headed to ultra-resolution? Do cell and molecular biologists need to go beyond super-resolution to get the information of value to them? Inevitably, there will be groups in the research community that will strive to achieve ever higher resolution, and others will find applications for it.
At present, observes Goodwin, resolution limits depend mainly on the size of autofluorescent proteins such as green fluorescent protein and labeled antibodies. Systems can get down to about the 15−20 nm range, about the size of the labeled antibody that is associated with the target protein of interest. “I think we will get to the resolution of individual molecules,” predicts Goodwin.
“I guarantee you that someone is working on [single] nanometer or picometer resolution,” adds Brian Manning, Ph.D., application scientist at Chroma.
As a manufacturer of optical filters and mirrors for microscope developers and end-users, Chroma is attuned to changing demands related to application trends. For example, there continues to be a lot of interest in optogenetics and the introduction of rhodopsin proteins into neurons to enabling imaging of individual neurons during live animal studies.
These types of experiments are part of an effort to map neuronal networks and map signaling pathways. He also describes demand from customers for increasingly flat dichroic mirrors, primarily for total internal reflection fluorescence microscopy applications, in which the flatness of the mirror is the limiting factor in being able to minimize deformation of the beam.
What Goes Around
“What’s old is new again,” says Manning, to describe the concept that the basic foundation of technology development involves a “constant revisiting” of what came before. A good example is the resurgence of Raman microscopy and specifically the technique commonly referred to as CARS, or coherent anti-stokes raman scattering, in which imaging is based on the vibrational signatures of molecules. This label-free method eliminates the need for fluorophores.
Label-free imaging techniques are increasingly being used to study neural architecture and to understand processes such as myelination and remylelination of neurons that have a role in neurodegenerative disease.
According to Brendan Brinkman, senior product manager for the scientific equipment group at Olympus America, using a technique such as CARS, researchers can “intrinsically image myelin” and, essentially, any lipids, making it a useful tool for studying plaque formation in atherosclerosis, for example, or in the development of drugs targeting obesity.
Olympus has employed a multimodal optical imaging approach that combines colocalized label-free imaging using CARS and two-photon microscopy for detecting protein autofluorescence and has demonstrated the ability to differentiate, for example, healthy arterial wall, early atherosclerotic lesions, and advanced atherosclerotic plaques.
For looking at live samples, “within the last year, we have seen advances in marker technologies that make it easier to image cells without damaging them,” says Paul Jantzen, marketing manager, core microscopy and research imaging at Olympus, “such as the smaller and brighter luciferase enzymes from Promega.”
As the markers are brighter and no fluorescence is involved, the cells are not subjected to high intensity light and imaging can extend over longer periods of time. Olympus plans to introduce its Luminoview LV200 luminescence microscope on the U.S. market before the end of this year to support luciferase-based cellular imaging.
The ability to do label-free imaging in whole tissue sections using two-photon confocal as well as conventional microscopy has been facilitated by the development of clearing, or clarifying agents that allow the light to penetrate deeper into the tissue sample for visualizing 3D structures.
“We can now do two-photon microscopy at the resolution of light microscopy and go down to 8 mm to look at intact tissues,” says Brinkman. The company’s Scaleview-A2 clearing solution and Scaleview immersion objectives were designed to facilitate imaging of large tissue areas at high resolution with the FV1200MPE system.
Not only are researchers better able to image large areas of fixed tissue in the x and y dimensions, but now in the z dimension too. “Whole slide imaging has really come to the forefront,” says Jantzen. “They can do it faster and are better able to manage the large amounts of data generated.”
For imaging multicellular constructs and tissues, two-photon microscopy allows for the use of longer wavelengths, which are less likely to interact with cells and be scattered, resulting in deeper light penetration and better z-resolution. These types of applications have also led to greater use of fluorophores that are excited by or emit far red or near infrared photons, according to James Joubert, application scientist at QImaging.
24 tiled areas composed of nearly 9,000 images stitched together using FluoView stage control and stitching software: 4 mm scaleview objective. [Olympus America]
Quantitation and Data Overload
Goodwin says he’s a firm believer in label-free techniques such as quantitative phase microscopy. He maintains that “this is up-and-coming technology” that requires much less light than fluorescent labeling techniques and allows for noninvasive monitoring of live cell dynamics.
Quantitation, in general, is a key trend in microscopy and will increase the ability to capture proteomic data.
Multiple factors have contributed to the emerging trend in quantitation in imaging, including improvements in assays, illumination sources, fluorochromes, and detectors. Quantitative output allows for comparison of results within and between laboratories, research groups, and collaborators within an organization and in different geographic regions. It also allows people without a microscope or imaging system to “look at” and gain information from images.
The main challenge in advancing quantitation is the need for greater standardization, in the view of T. Regan Baird, Ph.D., North American sales manager, microscopy division, at Lumencor. Instrument and software developers and users need to agree on a format and on whether and how to fit this new technology into existing standards or to modify the standards.
As an example, Dr. Baird points to the vast amount of data collected with new Scientific CMOS (sCMOS [Complementary Metal Oxide Semiconductor]) imaging technology, for which there is no standard format for databasing.
An example of the new sCMOS technology is Hamamatsu’s ORCA-Flash4.0 with a GEN II sCMOS detector.
To support imaging of large tissue samples and live cells and organisms, Duncan McMillan, director, product marketing, biosciences at Carl Zeiss, describes several new techniques and technologies that help overcome some of the key challenges.
For example, with a “smart microscopy” approach, users image a sample at a sufficient resolution to allow them to determine where they need to go back and visualize the sample at higher resolution to get the information they are seeking. This “first pass” approach is intended to avoid the problem of data overload and to focus on acquiring the most valuable and relevant data and accelerating the time to results.
At the recent Neuroscience meeting, Zeiss introduced the Lightsheet Z.1 LSFM imaging aystem, designed for 3D fluorescence imaging of large living specimens over hours to days with low photoxicity. The “light sheet” is an expanded light beam that illuminates a thin section of the sample; images are captured at a 90º angle to the light sheet, and images taken from different viewing angles can then be combined computationally into 3D reconstructions and time-lapse videos.
Also new from Zeiss is the Sigma VP 3View scanning electron microscope that incorporates an ultramicrotome within the SEM chamber to enable continuous cutting and imaging of thousands of serial samples from a fixed block of tissue to generate a 3D image at nanometer resolution.
McMillan also points to correlative microscopy techniques that facilitate this type of approach, and help researchers, for example, image a sample of live brain cells using a light microscope and then fix and label the sample for imaging in an electron microscope, with the ability to localize the image to the same site while accounting for factors such as tissue shrinkage or distortion.
Brinkman describes growing use of random access microscopy, a technique that involves moving from position to position within a sample and measuring fluctuations in fluoresence intensity, rather than scanning the entire sample. By targeting only positions of interest, data collection is faster, enabling speed relevant for physiological measurement in living tissues such as brain neuroanatomy.
The drive to increase the speed of imaging and to perform dynamic live-cell imaging is mainly limited by the capability of the detector to capture and transfer data and the amount of data storage capacity.
“You need to get the image data from the detector to the computer and stored,” says Dr. Baird, and a single image yields at least a half a megabyte of data, with thousands of images collected in a single experiment.
A number of different, synergistic advances are contributing to increased imaging speed including new light source technology, light engines that allow for rapid wavelength changing, synchronized with detectors that provide multicolor image acquisition while preserving the fluorophores, decreasing phototoxicity, and increasing signal-to-noise ratios.
Lumencor’s new SPECTRA X light engine™ is a hybrid solid state light engine that includes up to six sources with single band pass filters within the visible spectrum so users can select only the specific wavelengths of light they want to produce for multicolor fluorescence microscopy. In the future, Dr. Baird of Lumencor envisions more merging of technologies on multimodal platforms, such as an instrument that would combine fluorescence and electron microscopy capabilities.
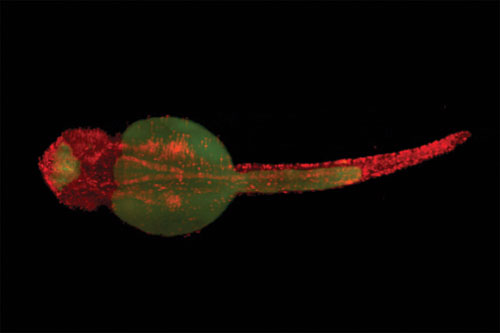
A two-day-old zebrafish labeled with H2A-mCherry (red) and autofluorescence (green). [Carl Zeiss]
Three Basic Pillars
The main trends going forward will focus on continued improvements in what Stanley Schwartz, senior advisor at Nikon Instruments, defines as “the basic three pillars of microscopy: speed, sensitivity, and resolution.”
This includes improving the spectral characteristics of the optics system to achieve broader spectral range at higher transmission rates, and improving or correcting spherical and chromatic aberrations. Schwartz identifies live-cell imaging as the largest market segment for research microscopy and cell biology.
“About 10 nm resolution is optimal size scale for cellular imaging,” says Steve Ross, general manager, products and marketing department at Nikon, and allows researchers to get down to the resolution of individual proteins. “Now we want to be able to do dynamic imaging,” i.e., to image over time in live cells, at that level of resolution.
One product targeted at live-cell imaging is Nikon’s Perfect Focus System designed to enhance long-term time-lapse imaging in live cells, providing the ability to remain focused by compensating for drift. The system incorporates a near-infrared 870-nanometer LED and CCD line sensor and offers a 5 millisecond (200 Hz) sampling rate, making it insensitive to rapid changes in focus such as may be caused by drug perfusion into the media.
“Super-resolution microscopy is having a huge impact on the market and will allow us to do correlative microscopy,” says Ross, adding that it will enable imaging at the molecular level with a specificity that is not achieved with electron microscopy. Software is becoming available that helps users correlate this new data with other imaging results generated using other microscopy techniques.
Schwartz and Ross envision continued strong growth in the area of stem cell research and medical applications of stem cell technology. Growing and studying stem cells presents particular challenges. Technology such as Nikon’s Biostations, including the Biostation CT and IM microscopy systems—contained incubation and microscope systems that enable time-lapse imaging of cells grown under control temperature, humidity, carbon dioxide conditions—allow for monitoring stem cell cultures without removing them from their environment to measure changes in growth, viability, or response to stimuli.
Live-cell dynamic imaging requires the ability to capture high speed events such as receptor binding and ion flux with sufficient spatial and temporal resolution. Existing technology can meet the need for imaging at 10−15 frames/second, but events such as ion signaling that occur at time scales of tens of milliseconds require high-resolution imaging at 100s of frames/second.
“And that need is not well met,” says Chris Ryan, a product development scientist at QImaging. sCMOS technology has the potential to overcome the combined challenges of high speed and resolution with reduced noise, but it remains a costly technology at present. This shortcoming is part of a larger issue in the microscopy field, which Ryan identifies as the total cost of ownership.
The imaging system comprises only a part of that total cost, which also includes high-performance computer capability, and the cable, computer card, and other devices needed to transfer and store the large amounts of imaging data generated. The raw speed and resolution for high-speed live-cell dynamic imaging can be achieved with sCMOS, and the goal now is to make the technology more broadly accessible.
Ryan explains that QImaging took a somewhat different approach with its first-generation sCMOS camera, the Rolera™ Bolt. Operating at a speed of 30 frames/second it was faster than a CCD camera and designed for live-cell imaging and motility studies in whole organisms, but was available at a fraction of the cost of higher speed sCMOS cameras.
Labeling technology is racing to keep up with advances in microscopy, says Joubert. In STORM, for example, a super-resolution technique that uses immunolabeling to image protein targets, the labeled antibodies are on the order of a few nanometers, which is about the same as the imaging resolution. Ries et al. published a method earlier this year for GFP-based super-resolution microsopy (Nature Methods) that used small antibodies called nanobodies linked to organic dyes to achieve nanometer spatial resolution with minimal labeling variation due to the size of the labeling agent and the ability to access structures unavailable to larger antibodies.
Merging Technologies
With newer technologies such as super-resolution, “as you are pushing the edge of the envelope, you need to use multiple techniques to confirm that your new findings are real,” says Joubert. “For example, if you are using super-resolution microscopy and trying to co-localize two molecules to see if they are interacting, you need to have supplementary techniques like FRET at the nanometer scale to confirm that they are interacting.”
Harald Fischer, marketing director at WITec, echoes the emerging trend among instrument manufacturers toward the integration of different microscope techniques in one system. WITec, for example, offers high-speed confocal Raman microscopes and integrated Raman-Atomic Force Microscope instruments. The new generation of confocal Raman microscopes enable “routine 3D chemical imaging, which was until then a point by point mapping rather than true confocal imaging,” says Fischer.
The company’s TrueSurface Microscopy Mode, an option available on the alpha300 microscope series that allows for topographic Raman imaging on large samples. “The functional core of the measurement mode is the sensor for optical profilometry, now fixed in the microscope objective turret,” Fischer says. “The system measures the surface topography of large samples and correlates it with confocal Raman microsopy,” without the need for extensive sample preparation.
When asked to describe three main trends in microscopy technology, McMillan, adds “ease of use and automation” to the two recurring themes of imaging large volumes of tissue at high resolution, and live-cell and organism imaging. This trend crosses technology and product boundaries—make microscope systems more user friendly.
A goal echoed by other companies and expressed by McMillan is to design and develop a unified user interface that can be used to control multiple different technology platforms, including devices from other manufacturers.
Niki Volkmann, marketing and project manager at Advanced Microscopy Group, recently acquired by Life Technologies, also identified “ease of use” as her number-one industry trend, followed by “green technology and price-to-performance ratios.” The company’s Evos® workstations exemplify those trends, according to Volkmann, and in particular, the newest addition to the Evos family, the FL Auto, an automated system with touch-screen technology and Wizard-based software.
Designed for cell culture applications, AMG’s XL Core microscope incorporates phase contrast and brightfield optics.
Instead of add-on modules and devices to enhance the capabilities of a standard compound microscope for specific applications, “companies are developing dedicated instruments that are designed to be user-friendly, turn-key systems with a low level learning curve,” says Vega.
For more on microscopes, be sure to check out our Expert Tips “4 Tips for Selecting the Right Microscopy System for You“.