March 15, 2010 (Vol. 30, No. 6)
Johann Maradey
Lisa Carey
Harry Glorikian Senior Executive, Board Director, Consultant, and Author
Segment Encompasses a Plethora of Critical Tools from Petri Dishes to Robotics for HCS
In the life science world, there has been a strong focus on next-generation sequencing and the $1,000 genome. The excitement in this nearly $30 billion life science tools industry should not be taken lightly; however, other segments of this market are also noteworthy. Cellular biology, for example, has demonstrated strong, sustained growth despite the sour economy.
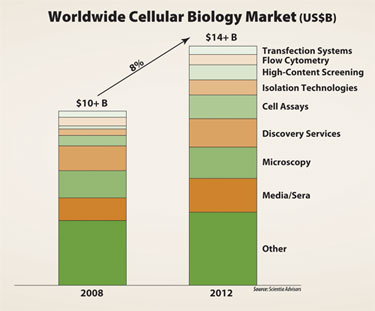
Cellular biology encompasses a plethora of research-focused technologies, from basic laboratory petri dishes to ultra-automated robotics for high-content screening. Cellular biology is currently the biggest revenue contributor to the life science tools industry, accounting for approximately 30% of total sales. It is expected to grow to a $14 billion market of its own by 2012.
Two major factors are driving growth in cellular biology: a recent surge in NIH/NSF funding and the ongoing need for biological relevance in drug discovery. The anticipated infusion of nearly $38 billion in funding for the NIH/NSF by the Obama administration this year is certain to keep academia busy and shareholders interested.
Despite a budget freeze for most nonmilitary discretionary activities in 2011, President Obama’s announcement of a $1 billion NIH budget increase indicates sustainable future growth for cellular biology.
In addition, the rapid discovery of cellular pathways and molecules is providing pharmaceutical and biotech firms with a large number of potential drug targets. Despite improvements in analysis technologies, however, the ability to thoroughly understand a potential drug’s biological profile remains difficult.
As a result, predicting the effects of drug candidates early in the pipeline is problematic. In fact, most existing drugs today exhibit clinically unforeseen, albeit harmless, pharmacological profiles that were unobserved until advanced stages of drug development or, in some cases, after commercialization. Whenever these side effects prove to be harmful, pharmaceutical companies lose significant market share.
With increasing drug development costs, researchers are seeking ways to eliminate candidates with dangerous toxicity and metabolic profiles early on. For example, unforeseen metabolism issues not determined in cell and rodent models account for 30% of all Phase I trial failures. The promise of coupling the existing target-based drug discovery model with a cell and systems biology approach promises to decrease the number of failures and lower costs early in the drug discovery pipeline.
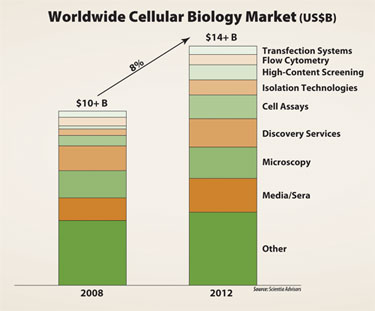
Cellular biology is currently the biggest revenue contributor to the life science tools industry.
Cell-Based Assays
A promising area within cellular biology is cell-based assays. With a compound annual growth rate of 16% from 2008 to 2012, these assays are a prime example of how the need for biological relevance is driving growth. Cell-based assays are procedures used to analyze and understand how live cells respond to external factors. Usage includes: drug discovery for validating drug targets and lead profiling; basic research for understanding cell signaling and function; and absorption, distribution, metabolism, and excretion (ADME) testing.
Traditional drug discovery assays have been target-based, designed to measure the effect of a compound on a particular cellular entity. However, research and drug discovery trends are changing. Researchers in academia and industry want to see the effects of their compounds on the whole cellular system, in turn creating opportunities for more therapeutic options.
Phenotypic assays have arisen as a potential solution to this problem. In addition to target-based assays, researchers have begun running high-content target-blind screens that provide system-wide information on the effects of compounds. Despite the amount of information gleaned from phenotypic assays, target-based assays will remain a necessity for understanding the pharmacological profile of compounds.
Recent improvements in high-content screening and data analysis are enabling the increased use of phenotypic assays. As a result, opportunities exist for companies that have competencies in reagent development and cells for creating novel ways of studying cellular systems.
Emerging Economies
Large investments by pharmaceutical firms in emerging economies are also driving growth in cell biology. Asia, in particular China, is the leading target for these investments and, if the trend continues, is expected to surpass Europe in R&D intensity within the next three to four years. An example of such an investment is AstraZeneca’s $100 million facility in Shanghai, which is focused on R&D for cancer therapies. Similarly in 2008, GlaxoSmithKline invested $116 million in an Asia-based R&D facility in Jurong, Singapore.
The increasing number of clinical research organizations (CROs) in China and India is another driver of cellular biology sales to emerging economies. Within the last five years, the number of CROs in China has grown rapidly, increasing service offerings and consequently driving outsourcing.
Chinese companies are taking advantage of the outsourcing opportunities, expanding from basic chemistry offerings to include lead optimization, enzyme/cell assays, and ADME/Tox studies. This extensive menu of services, combined with low-costs and innovation, is positioning China as an R&D services competitor to the U.S.
Cellular biology is a segment of the life science tools industry that should not be overlooked. Driven by government funding, a changing pharmaceutical R&D philosophy, and increasing R&D presence in emerging economies, cellular biology and its encompassing technologies have shown impressive resilience in the midst of a frightening economic environment.
Johann Maradey and Lisa Carey are analysts, and Harry Glorikian ([email protected]) is managing partner at Scientia Advisors.
Web: www.scientiaadv.com.