December 1, 2006 (Vol. 26, No. 21)
Proper Identification and Implementation Can Help Users Avoid Common Pitfalls
Water is probably the most commonly used laboratory reagent. However, it is often taken for granted and its potential impact on experimental outcomes overlooked. Understanding basic principles regarding laboratory water may help scientists avoid common pitfalls. Millipore (www.millipore.com) has extensive experience with water purification systems as well as water quality monitoring based on customer interaction.
Identify Water Quality Needed
Tap water commonly contains substances that may be classified as particles, ions/inorganic compounds, organic molecules, microorganisms, and dissolved gases. Depending on how the water is used in the laboratory, some of these contaminants may or may not have an influence on experimental results.
For example, in liquid chromatography, the water used in the mobile phase should be devoid of organic molecules for good quality baselines. However, the presence of organic molecules in the water would have less impact on applications such as glassware rinsing, autoclaving, or microbiology. Before setting up a new laboratory or even starting a new experiment, it is important to identify the specific water quality needed.
Various organizations and regulatory bodies have defined water quality standards. As they all differ slightly it is difficult to define clearly the types of laboratory-grade water available. The most common classifications are:
• Type I. This water (also known as ultrapure) contains extremely low levels of ions (resistivity 18.2 MW.cm), organic molecules, bacteria, and particles. Usually, it is produced by combining purification technologies such as ion-exchange resins, activated carbon, reverse osmosis, ultraviolet photo-oxidation, filtration processes, and electrodeionization. It is used not only for analytical methods such as high-pressure liquid chromatography (HPLC), gas chromatography, and inductively-coupled plasma mass spectrometry (ICP-MS), but also to prepare reagents for molecular biology and cell culture.
• Type II. This water (also known as pure) may contain a small amount of ions (resistivity above 5 MW.cm), organic molecules (up to 50 ppb of total organic carbon), and possibly a small amount of bacteria. It is used commonly in preparing everyday reagents and buffers.
• Type III. This water usually is generated by reverse-osmosis or ion-exchange. It is used for less sensitive applications such as qualitative analyses, glassware rinsing, and water baths.
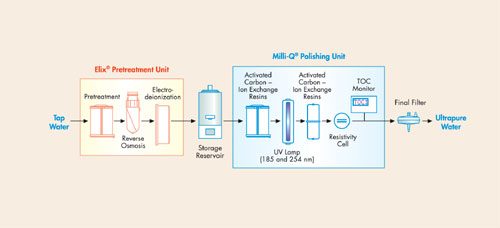
Based on their specific areas of activity, laboratories may use one or more of these types of water. Figure 1 illustrates the combination of purification technologies used in an ultrapure water purification system.
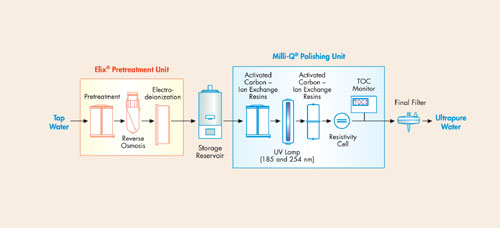
Figure 1
Choose a Water Purification System
After defining the type(s) of water needed, the next step is to select or design a water purification system tailored to the laboratory needs. It is particularly critical to assess the required daily/weekly water volume as accurately as possible. Optimally, purified water should be available when needed with minimal storage time. In addition, consider the space requirements. Purification systems now can be installed under the bench or wall-mounted to preserve work space. The need for pure and ultrapure water on demand in different laboratory locations or in a set of laboratories is an important criterion when selecting the water purification solution. A total water solution approach is beneficial when building new facilities in order to integrate all these parameters in the water purification system design.
Importance of Water Pretreatment
Many research facilities or universities have an “in-house” supply of deionized water. The potential difficulty is the reliance on others to maintain the system and change the ion-exchange resins on time. Exhausted resins may leach high levels of contaminants into the water all at once. In addition, repeated resin regeneration cycles lead to broken resin beads as well as the release of particles and organic molecules into the water. This also is conducive to bacterial growth.
Even if this water is further purified with a polishing system, the large variations in water quality at the pretreatment level generate inconsistent and poor quality ultrapure water. Therefore, the pretreatment step is a key step in the purification process. Pretreatment systems that include a combination of purification technologies such as ion-exchange resins, activated carbon, reverse osmosis, and electrodeionization have been shown to produce consistent and reliable water quality. They also optimize the performance of further purification steps.
Avoid Storing Purified Water
High purity water has a high affinity for chemicals in its surroundings. It can absorb chemicals readily from storage containers and pipes as well as chemical vapors from the laboratory air.
The polyethylene carboys and wash bottles commonly used to store water in laboratories can leach organic molecules into ultrapure water. Figure 2 illustrates how the HPLC baseline quality degrades quickly with time when ultrapure water is stored in a polyethylene container. Trace enrichment was performed by running 60 mL of each sample on a C18 column, followed by a linear gradient run (95% water to 100% acetonitrile in 30 min).
Glass bottles also may leach inorganic compounds. Therefore, it is recommended to use ultrapure water immediately after production. One benefit of a purification system is the ability to get ultrapure water on demand.
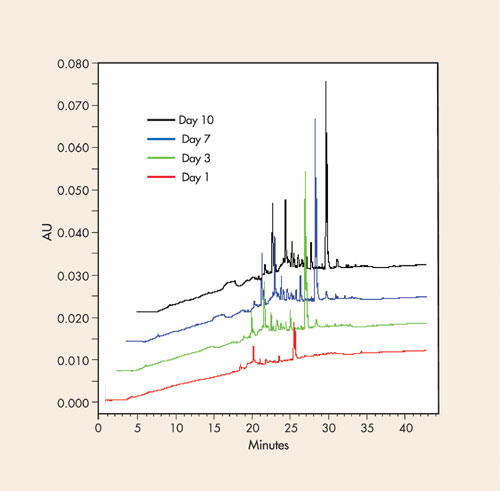
Figure 2
Monitor Water Quality
Monitoring water quality ensures experiment repeatability and quality results. Commonly used monitoring tools are conductivity and total organic carbon (TOC) meters. Conductivity measures the flow of electrons through a fluid, which is proportional to the concentration of ions, their charge and mobility. Therefore, conductivity (or its reciprocal, resistivity) is a good indicator of overall ionic water purity.
The theoretical conductivity of pure water is 0.055 µS/cm at 25°C and the corresponding resistivity is 18.2 MW.cm. TOC analysis indicates the total amount of organic material present in water. It is commonly measured by converting organic matter into carbon dioxide using ultraviolet photo-oxidation, or chemical oxidation, then measuring the change in water conductivity.
Assuming that a resistivity reading of 18.2 MW.cm is indicative of ultrapure water is inaccurate as it only indicates that no ionic species are present in water. Many organic molecules do not ionize readily and, as a result, do not influence the conductivity of water. Therefore, purified water may contain some organic compounds and have an excellent resistivity reading.
Similarly, a low TOC value only indicates that there are no organic molecules present in water without indicating the levels of metals or salts. Combining these two monitoring tools reflects the level of water purity more precisely.
Avoiding Bacteria
Some contaminants not accounted for by water purification monitoring systems may also affect experimental outcomes. Bacteria are the most overlooked. Once started, bacterial contamination may propagate rapidly throughout the system. Bacteria eventually form biofilms, which are difficult to remove. They release small organic molecules and inorganic ions into the water, which may appear as extraneous peaks on ion chromatography and HPLC chromatograms.
As particulates, bacteria present in water can increase column back pressure in liquid chromatography or spoil ICP nebulizers. Bacteria may release a number of macromolecules, such as enzymes (e.g., nucleases, alkaline phosphatase) and endotoxins.
Nucleases have a detrimental effect on molecular biology experiments based on the use of nucleic acids (RNA and DNA). Bacterial alkaline phosphatase may interfere with enzyme immunoassays using calf intestine alkaline phosphatase as a label.
Endotoxins interfere with cell function and growth in mammalian cell cultures. These macromolecules can be monitored using biochemical kits and removed using ultrafiltration. An ultrafiltration unit can be installed either inside the purification system or at the point-of-use. Bacterial development inside water purification systems can be minimized by not leaving water stagnant in reservoirs or in the purification system, performing routine maintenance, and changing the cartridges regularly.
One Size Does Not Fit All
It might be tempting to select ultrapure water for all the applications in the laboratory. However, in some cases, not only would ultrapure water be unnecessary and expensive, but also it may lead to poorer results. These instances include some specific biological applications where trace elements may be beneficial.
One example is the water used to raise experimental zebrafish. These fish require water without chlorine, pesticides, ammonia, and nitrite. They are also sensitive to pH variations. Ultrapure water has a pH of 7.0. However, when exposed to the air, it readily absorbs carbon dioxide, which forms carbonic acid and lowers the pH to approximately 5.6. These pH variations would be detrimental to the fish. Although the system water commonly is prepared by adding a salt mixture to the water, this mixture does not contain all the trace nutrients necessary to the good health of the fish. Good quality reverse osmosis water would be preferred to ultrapure water, devoid of these important trace minerals.
Other situations where Type II or III water may be preferred to Type I are related to the aggressive nature of ultrapure water. For example, ultrasonic baths or other systems where the water is in prolonged contact with metals may eventually be damaged by ultrapure water. Therefore, it is important to tailor the type of water used to each specific use.
It has been many years since deionized and distilled water were the only purified water choices available. Today, various combinations of purification technologies are used to remove contaminants efficiently. Therefore, matching the water quality with each application yields more consistent and reliable results. Scientists should partner with companies to identify and install the water systems that meet their scientific and space concerns.
Estelle L. Riche, Ph.D., is applications support scientist and Stephane Mabic, Ph.D., is worldwide application support manager, R&D in the Bioscience division, for Millipore. Web: www.millipore.com. Phone: +33 13012 7140. E-mail: [email protected].