October 15, 2009 (Vol. 29, No. 18)
Nadia A. Badr
Brad Justice
Robin A. Felder, Ph.D.
Microcarrier System Designed to Keep Up with High-Content Screening Demand
In recent years, there has been a significant decline in the success rate of drug candidates in clinical trials. This situation has put pharmaceutical companies under immense pressure to reduce expenses and increase productivity. As a result, drug researchers have stepped up their use of high-content screening of large chemical libraries. Larger screens, however, do not always produce more leads.
Use of target cells in culture that more closely resembles the target organ in vivo can increase predictability. In addition, the increased biological relevance of cell lines and their drug targets decreases late attrition in trials.
At the same time, an increase in the use of cell-based therapeutic strategies has escalated the need for cells that are of consistent quality and in sufficient quantities. Automated platforms are being used to meet the increased demand for in vivo mimetic organotypic cells. The ability to grow large numbers of cells in a consistent and reliable fashion is directly related to the automation of the cell culture process, sample handling, imaging, and assaying.
Over the last 30 years, the drug screening industry has seen a trend toward using more biologically relevant 3-D surfaces for cell-based screening. Cells interact with neighboring cells and the extracellular matrix (ECM) in vivo by means of biochemical and mechanical cues. Cell cultures that replicate real tissue interactions are better candidates for proliferation, migration, apoptosis, and tumorigenesis models.
Currently, the vast majority of cell culture for cell-based data harvesting occurs in flat, hard, plastic dishes. It is widely accepted that this system has limitations in biological relevance, thus, a number of new scaffolds have been developed to mimic the ECM in vivo environment. Recent research has demonstrated that coating flat plasticware with reconstituted basement membrane (rBM) provides additional insights into the extracellular context in tumorigenesis and reveals differences in receptor activity in human breast cancer cells. Though such scaffolds have revealed much about cell behavior, microcarriers take 3-D biology to the next level.
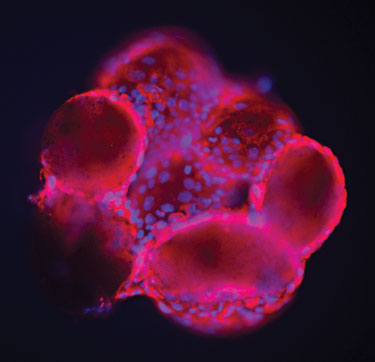
Figure 1. Human embryonic stem cells (HU-ES 16) are cultured on laminin-coated GEM. The cells are stained with alkaline phosphatase (red) and DAPI (blue). (Image and data courtesy of StemGent.)
Microcarriers
Microcarriers are small spheres that can range in diameter from that of the cell up to 500 µm. Their many benefits include supporting the growth of difficult or sensitive cell types. The ideal microcarrier is made of biologically relevant and in vivo mimetic constituents.
Figure 1 shows human embryonic stem cells cultured on the GEM™, a microcarrier developed by Global Cell Solutions (GCS). GEM consists of an alginate core surrounded by a covalently bound molecular layer of protein. Cells cultured on GEM demonstrate more in vivo-like characteristics than those grown in 2-D flasks.
Alginate is a particularly biologically relevant material because it has structural constituents that resemble the cell-supporting network in vivo. The molecular structure of alginate is similar to that of hyaluronic acid, which is a constituent of the extracellular matrix and has been shown to be beneficial in keeping stem cells undifferentiated. The unique porous matrix stimulates the growth of cells and allows for polarized ion transport mimicking the in vivo metabolic state. Alginate is approved by the FDA for use in humans, making it an ideal candidate for future cell-based therapeutics.
Furthermore, a variety of protein layers such as collagen or laminin can be coated onto the alginate to accommodate the basement membrane needs of different types of cells. Human embryonic stem cells, for example, can be cultured feeder-free and expanded on purified laminin-coated GEM and retain embryonic stem cell markers as in Figure 1.
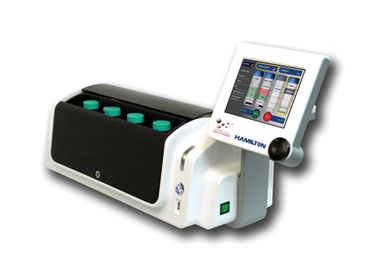
Figure 2. The BioLevitator R3 is a benchtop incubator and bioreactor capable of maintaining four high-density 3-D microcarrier cultures.
Scaling Up Cell Culture
Microcarrier culture is recommended for high-density cultures—in some cases, densities as high as two million cells per milliliter can be realized. Microcarrier technology lends itself to large-scale production because the frequent media changes and cell dispensing in downstream processes (such as cryopreservation) can be fully automated. The magnetic property of GEM allows the automation system to either disperse or aggregate the substrate at any stage of the process.
The BioLevitator™ is a benchtop incubator and bioreactor produced for GCS by Hamilton for cell culture on the GEM substrate. It uses programmable magnets to selectively manipulate the position and motion of the cell-coated GEM. The BioLevitator R3 (Figure 2) has touchscreen control and internal carbon dioxide and temperature regulation and can be integrated into most liquid-handling platforms.
Automated cellular systems allow rapid visualization and analysis of large groups of cells. With GEM, cells can be automatically placed directly into the assay process without removing them from the growth surface. The GEM is optically clear and nonautofluorescent allowing cells to be assayed for absorbance, fluorescence, and luminescence, as well as imaged directly.
Microcarriers as substrates in cell culture can provide a significant cost reduction. In a recent study, labor and waste expenses (measured by disposable plastic and glass biohazard waste) were reduced by 85% and 76%, respectively (Figure 3).
GCS and Hamilton are developing a high-throughput system that will automate cell culture and cell passaging, integrating up to four BioLevitators on the deck of a Microlab® STAR. This 3D CellHOST™ system will enable large-scale growth and expansion of cells using the magnetic microcarrier.
Three-dimensional microcarrier cell culture provides a solution to the current constraints in cell procurement. Specifically, substrate characteristics that include magnetism, extracellular matrix-like constituents, and the ability to be pipetted in liquid phase, all contribute to a highly automatable cell culture system that can keep up with modern high-content screening demands.
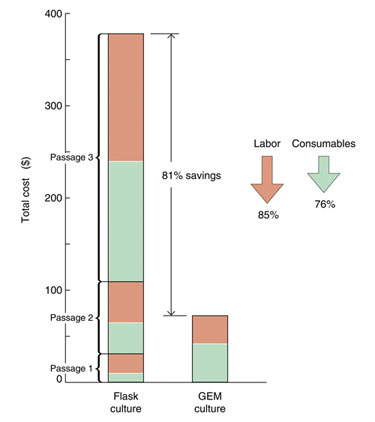
Figure 3. Process cost analysis is compared between conventional cell culture and 3-D cell culture on GEMs.
Nadia A. Badr ([email protected]) is senior scientist, and Brad Justice is vp of R&D at Global Cell Solutions. Web: www.globalcellsolutions.com. Robin A. Felder, Ph.D., is professor of pathology at the University of Virginia.







