December 1, 2010 (Vol. 30, No. 21)
Sue Pearson Ph.D. Freelance Writer GEN
Methodologies Like SPR and ITC Are Saving Researchers Time and Money
There is an explosion of biophysical methods being used in the discovery phase of biotherapeutics, according to Fredrik Sundberg, director of strategic market development at GE Healthcare.
“Label-free technologies including surface plasmon resonance (SPR) and isothermal titration calorimetry (ITC) are becoming more widespread. This is because scientists are more aware of how to interpret the data from these techniques, and are using them for designing better biological drugs and vaccines. Also, these are real-time assays, which can offer significant time savings,” Sundberg explained at the recent “Developments in Protein Interaction Analysis” (DiPIA) meeting in Barcelona.
Noxxon Pharma is just one of the many companies using label-free technology for discovery and preclinical development. “Aptamers are oligonucleotides that bind and efficiently inhibit target molecules in a manner conceptually similar to antibodies,” reported Christian Maasch, Ph.D., director of biophysical analysis.
“They are identified by an in vitro selection process. Conventional RNA or DNA aptamers are susceptible to degradation by ubiquitous nucleases. A stabilized aptamer, Eyetech’s Macugen, has been approved by the FDA as an intravitreal injection for the treatment of age-related macular degeneration.
“Noxxon has developed chemical entities based on mirror-image RNA oligonucleotides, which we call Spiegelmers®. These don’t occur in nature, are not metabolized, and do not hybridize with native nucleic acids.
“We are using SPR to support lead candidate identification and preclinical development, including hit analysis, kinetic evaluation, selectivity determination, and epitope mapping to characterize their binding activities, search for potential antidrug antibodies, and detect plasma protein binding.”
Using these applications of SPR the company has identified two lead candidates, NOX-E36 and NOX-A12, that have been tested in Phase I trials. NOX E36 (MCP 1 antagonist) is going to be developed to treat diabetic complications such as nephropathy. NOX A12 (SDF 1 antagonist) can be used to mobilize hematopoietic stem cells from bone marrow and to treat hematological and solid tumors.
“We believe the Spiegelmer technology combines the benefits of small molecule drugs and biopharmaceuticals, including the easily scalable chemical manufacturing process, the exceptional target specificity, and inhibitory potency. In preclinical, as well as clinical studies, we have confirmed that the beauty of Spiegelmers, besides their exceptional stability, is that they don’t activate the innate immune responses like conventional oligos can and are eliminated by the kidneys as intact product with a half-life of more than 40 hours. The recent progress in the clinics shows that Spiegelmers are well suited for further drug development, which is planned to be partnered at later stages.”
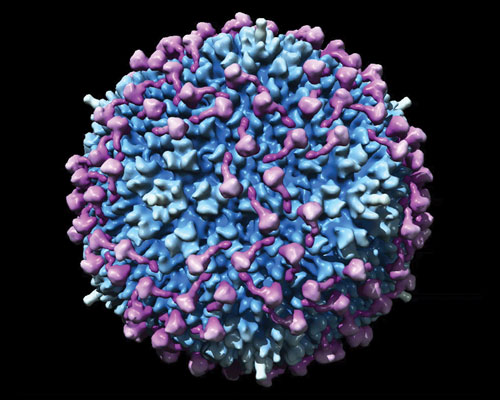
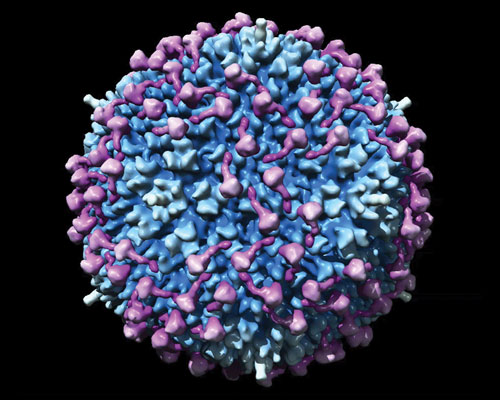
Molecular model of coagulation factor X binding to adenovirus type 5 (based on cryoelectron microscopy data): Adenovirus is in blue and factor X in purple. [GE Healthcare]
High-Specificity Antibodies
Another application where label-free technologies helped to determine structural binding elements of a Fab fragment was presented at DiPIA by Rajkumar Ganesan, Ph.D., a postdoctoral fellow in Daniel Kirchhofer, Ph.D.’s lab at Genentech.
Dr. Kirchhofer and his colleagues are working on understanding the structural and mechanistic aspects of serine protease inhibition by antibodies targeting the active or allosteric site. “When transmembrane serine proteases such as hepsin are overexpressed in preclinical prostate cancer models they promote tumors; blocking of the active sites of hepsin with an antibody could help inhibit tumor growth,” Dr. Ganesan explained.
“However, we could not find inhibitory antibodies using classical hybridoma techniques or phage-display libraries. But using an active site blocked version of hepsin and a phage-display library of restricted chemical diversity at the complementarity determining regions, we found a number of Fabs that did bind to hepsin.”
Dr. Ganesan presented SPR data showing that one of these Fab fragments, Fab25, has high affinity binding for hepsin, and its inhibitory activity was confirmed by enzyme assays using a synthetic substrate. SPR was also used in competition binding experiments with two active site derived inhibitors and demonstrated that Fab25’s binding site has structural elements at or around the hepsin active site region. Using ITC, Dr. Ganesan showed that Fab25’s binding is enthalpically driven, indicating potential hydrogen bond formation and electrostatic interactions between Fab25 and hepsin.
“Our Fab25 fragment is a potent and specific inhibitor of hepsin and may be a good tool for inhibiting hepsin in prostate cancer.”
In vaccine design, the use of SPR is spreading because it can be used for specificity testing, epitope mapping, and immune response ranking. “Many of the major pharmas are using SPR to check their viral vaccines because it is now possible to qualify the virus from culture supernatant,” Sundberg explained. “For example, there are vaccine companies using SPR to verify the existence of antigenic sites on virus like particles with specific antibodies.
“Trends in vaccine development are toward more complex vaccines and lower dose vaccines. These require more flexible, sensitive methods of measuring vaccine concentration, and SPR can achieve this.”
There is a strong push in viral vaccine production to move from egg- to cell culture-based production, and there is growing interest in using SPR for process development. Additionally, because SPR is being successfully used at the research stage for checking vaccine potency, it could also be used as a manufacturing method for batch-release verification.
Sundberg presented results of a study where scientists at GE Healthcare compared the current gold standard method for batch release of flu vaccine, the single radial immunodiffusion (SRD) assay, with an SPR method. In the inhibition-based SPR assay, three different virus hemagglutinins (HAs) were immobilized on a Sensor CM5 in a Biacore™ system. Virus from the sample was then mixed with a fixed concentration of anti-serum from the National Institute for Biological Standards and Control and injected over the surface.
The results showed that using the SRD assay, 5 µg/mL could be measured, whereas the more sensitive SPR assay measured 0.5 µg/m for all three subtypes. “One major advantage of using the SPR assay is that it is less labor intensive,” Sundberg explained.
“The potency of the three antigens in the flu vaccine can be measured simultaneously, but using the SRD assay the potency of each antigen has to be calculated separately by measuring inhibition zones on three different gels. Since SPR is more sensitive it would be valuable in influenza purification process development, where fractions from optimization experiments tend to have low concentrations of virus. There may also be an opportunity to fill each vaccine vial more accurately and potentially make significant cost savings.”
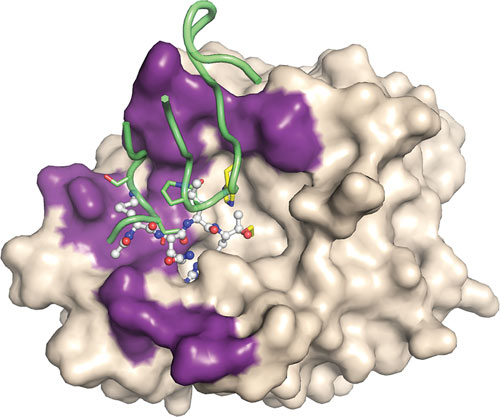
Inhibitory antibody targeting the serine protease active site: Superposition of the Fab58/HGFA structure (PDB code 2R0K) with the structure of HGFA/peptidic inhibitor (ball and stick) complex (PDB code 2WUC) to illustrate the HGFA protease (surface model–beige) subsite occupancy by Fab58 CDR-loop residues (green). The antibody binding region on HGFA is highlighted in purple. [Genentech]
Safer Delivery
John McVey, Ph.D., head of experimental and translational research at the Thrombosis Research Institute, showed how using SPR data has helped in re-designing an adenovirus vector. According to Dr. McVey, the adenovirus is the most commonly used vector in gene-therapy trials, yet very little is known about its physiology. The majority of the vector is transferred into the liver, which can lead to toxicity problems, and it is this lack of understanding that led Dr. McVey and his team to study the virus.
Dr. McVey presented SPR data showing how a blood coagulation factor (FX) binds to the hexon on the virus surface and using electron cryomicroscopy they demonstrated the FX bound to hypervariable regions (HVRs) in the hexon protein. A series of recombinant adenovirus vectors was engineered in which the HVRs were swapped and using SPR these vectors were shown not to bind to FX. The recombinant vectors were further altered by site-directed mutagenesis in the HVRs, and using SPR the amino acids in the HVRs that bound to FX were identified.
“Using SPR we now understand a little more about how the virus could be binding to hepatocytes, we could generate new types of adenovirus, which can bypass the liver. This means we may be able to improve the safety profile of gene therapies using these new kinds of vector.”
The Future
Delegates and speakers at the DiPIA meeting agreed that the use of label-free approaches is now more frequent in discovery of biotherapies and vaccines. However, techniques such as SPR have also shown promise in improving many downstream analytical tests, including ADME/Tox screening, bioprocessing, and QC applications.
According to Dr. Maasch, Noxxon plans “to use SPR later in development work such as pharmacokinetic analysis using an anti-PEG antibody to track Spiegelmers. As Spiegelmers are pegylated, this will require less assay development time if we use SPR.”
Many academic groups have been able to link bioreactors to SPR methods to provide real-time analysis of mAb production and this could be used in pharmaceutical manufacturing.
“To ensure that Biacore instruments could make the transition from being analytical lab machines to being used in a GMP environment, we have a clear link between paper and electronic records. We also have trained personnel that will help to document everything—because the FDA’s view is unless it’s documented it never happened. SPR assays are already being used in batch-release applications by some biopharma companies. We expect that over the next few years other companies will follow and start implementation of label-free assays in this key area,” Sundberg said.
SPR assays are already being used in batch-release applications by some biopharma companies. We expect that over the next few years other companies will follow and start implementation of label-free assays in this key area,” Sundberg added.
In short, label-free technologies are going from strong to stronger in the development and manufacture of biotherapies and vaccines. “When the pharmas and biotechs start trying to gain approval for their biosimilar products, multiple independent technologies such as SPR, ITC, ELISA, and LC/MS, will provide a sea of protein data.
“If the biosimilar is very well characterized and its protein profile compares well with the existing antibody or protein therapy, then regulatory authorities may allow the company to perform smaller numbers of clinical trials to get their biotherapeutics to market. This could significantly reduce the clinical development costs of the therapy and is a good reason to characterize a biosimilar very thoroughly,” Sundberg concluded.