September 15, 2011 (Vol. 31, No. 16)
K. John John Morrow Jr. Ph.D. President Newport Biotech
In Vivo-Like Environment Paves Way toward Fresh Insights and Whole-Organ Generation
In vitro cell culture has been a powerful model for in vivo biological processes. However, the majority of studies have been conducted using 2-D culture systems in which cells grow as flat monolayers. Two recent symposia—CHI’s “Bioprocessing Summit” and the “BioMethods Boston” conference—discussed a raft of studies on cellular behavior in 3-D platforms, and how this model may offer a more realistic view of how cells behave in their natural environments.
“Deregulated cell movement is a hallmark of pathological events, including pulmonary disease and cancer metastasis,” said Shuichi Takayama, Ph.D., professor of biomedical engineering at the University of Michigan. To investigate this phenomenon, Dr. Takayama and his team developed a high-throughput platform for cell-migration studies.
Traditionally, cell migration could be measured by the wound-healing assay, in which a cell monolayer is scraped with a tool. This leaves a vacant area, which in subsequent days is repaired as cells move to fill in the empty zone.
This approach is not entirely satisfactory, as there is a need for specialized tools to scrape the cell monolayer in high-throughput settings, and the scratching tool may disrupt both cells and the underlying substrate. Moreover, since the cell removal may be initiated by hand, the “wound” size (or width) may be variable, yielding inconsistent results in duplicate assays.
To meet these challenges, Dr. Takayama and his colleagues took advantage of an aqueous two-phase system consisting of polyethylene glycol (PEG) and dextran (DEX) as the phase-forming polymers.
A submicroliter droplet of the dextran phase is spotted and dried in a conventional microwell. Addition of the polyethylene glycol to the well results in the rehydration of the dextran spot to form an immiscible droplet.
The PEG-DEX biphasic system generates an interfacial tension, which prevents cells from adhering to the part of the substrate covered by the rehydrating drop. The result is a consistent, repeatable, clearly defined circular area from which the cells are cleared away, Dr. Takayama claimed.
He and his team have investigated another model for cell culture evaluation: spheroid formation. Using custom-designed 384-array plates, a pipette is used to introduce the cells into holes where the droplets are suspended. The pipette tip is first inserted through the access hole to the bottom surface of the plate, and the cell suspension is dispensed.
The cell suspension is attracted to the hydrophilic plate surface and a hanging drop is quickly formed and confined within the plateau. Within hours, individual cells will begin to aggregate and form into a single spheroid. The hanging drops can be fed and manipulated on a periodic basis. Dr. Takayama is developing this project in collaboration with 3D Biomatrix.
Breast Cancer Tumorigenesis
“Increased stiffness is a hallmark of solid tumors, due to the altered physiochemical properties of the extracellular matrix,” stated Claudia Fischbach-Teschl, Ph.D., assistant professor of biomedical engineering at Cornell University. “This is thought to be largely due to crosslinking of collagen, which occurs with greater incidence than in normal breast tissue.”
Other elements may participate in this process including fibronectin, whose stretching leads to unfolding and increased stiffness of the molecules. Furthermore, these modifications of the fibronectin matrix may contribute to the enhanced rigidity of mammary tumors, which is associated with tumorigenesis and metastasis.
Dr. Fischbach-Teschl, in collaboration with Delphine Gourdon, Ph.D., a researcher in the department of material science and engineering at Cornell University, have evaluated paracrine signaling between breast cancer cells and adipose progenitor cells in order to assess whether such signaling might promote tumor progression via changes in fibronectin matrix assembly.
They constructed in vitro and in vivo model systems combining adipose cells with a human breast cancer cell line. In order to measure, quantitatively, the changes in the fibronectin molecule, they employed FRET analysis.
Results demonstrated that the paracrine signaling between the tumor cells and the adipose cells contributes to fibronectin matrix stiffening in tumors. Furthermore, they showed that this stiffening is leading to phenotypic changes of adipose stem cells that lead to enhanced tumor progression.
“Our observations suggest that these changes promote tumor vascularization and growth by upregulating the pro-angiogenic capability of both the adipose stem cells and the endothelial cells,” Dr. Fischbach-Teschl explained. “In summary, these studies suggest that adipose stem cells stimulate tumor vascularization in a stiffness-dependent manner and represent a promising target for improved anti-angiogenic therapies.”
Engineering a Transplantable Liver
Basak Uygun, Ph.D., an instructor at the Center for Engineering and Medicine at Massachusetts General Hospital, leads a project aimed at developing transplantable liver grafts. This approach involves the portal perfusion of rat livers with SDS, an anionic detergent that lyses cells and solubilizes cytoplasmic components, leaving a translucent scaffold, which retained the shape of the liver.
While no viable cells remain, the matrix consists of a variety of extracellular matrix proteins, including collagen and glycosaminoglycans. The scaffold contains an intact microvasculature and can serve as a construct for rebuilding an artificial liver.
Dr. Uygun’s team determined that the scaffolds can be reseeded with primary rat hepatocytes at high efficiency; they used as few as 50 million cells (or 5% of the normal liver population), which was previously shown to be sufficient to restore liver function in the rat model. This number could be further increased up to 200 million or 20% of the original mass.
Hepatocyte viability and metabolic function were maintained in perfusion culture, including albumin production and gene expression equivalent to 20% and 30% of in vivo levels, respectively.
Previous attempts have failed in creating complex tissue-engineered constructs for liver transplantation, Dr. Uygun said. While these latest results are quite encouraging, she cautioned that there are still many problems that need to be resolved before a fully functional artificial liver is available.
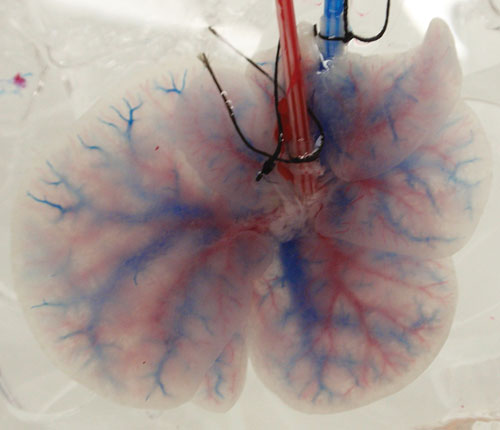
Scientists at Massachusetts General Hospital are developing transplantable liver grafts. Decellularized liver retains both venous (blue) and portal (red) network of blood vessels.
Challenges in Creating Whole Organs
Despite years of investigation, the successful development of 3-D matrix scaffolds that will successfully support a recellularized, functional organ has many barriers to overcome, according to Stephen Badylak, M.D., Ph.D., a professor in the department of surgery at the University of Pittsburgh.
Dr. Badylak believes that the extracellular matrix (ECM) represents an ideal support scaffold for regenerative medicine applications. “Recent advancements in decellularization and recellularization techniques and promising preclinical studies suggest that organ engineering is a very real possibility in the foreseeable future,” he stated.
Yet Dr. Badylak enumerated many formidable challenges, including the appropriate choice of candidate species from which the donor organ can be harvested, optimal methods of removing and reintroducing cells into the ECM, and selection of the most appropriate cell populations.
He cautioned that none of the whole-organ grafts produced to date have been used to replace or support function in vivo for more than a few hours, aside from skin grafts.
The formation of fibrotic tissue and scarring is a significant problem in the engineering of artificial organs. In the case of skin, in which vascularization has not sufficiently occurred, extensive scarring can profoundly compromise the quality of life for patients.
For this reason, Dr. Badylak suggests that protocols in which vascularization is promoted would improve the outlook for all types of whole-organ engineering. Sounding a cautionary note, Dr. Badylak stated, “it is important not to claim victory prematurely and create overly optimistic expectations until indisputable success in animal models of organ failure is demonstrated.”
Magnetic Levitation
A new approach to assembling 3-D cell cultures is being pursued by Thomas Killian, Ph.D., and his collaborators at Rice University and Nano3D Biosciences. “Magnetic levitation through the use of magnetic nanoparticles is a new paradigm, providing the advantages of a simple platform that can easily be incorporated into existing protocols and diagnostics,” Dr. Killian asserted.
While there are a number of studies under way using biodegradable porous scaffolds, protein matrices, protein-based gels, or rotational-based bioreactors as a basis for three-dimensional in vitro structures, Dr. Killian argued that there are significant advantages to his approach.
He described a three-dimensional Bio-Assembler™ that relies on cellular uptake of a biocompatible gel containing magnetic iron oxide and gold nanoparticles.
Dr. Killian pointed out that the biological application of magnetic forces is well recognized. Magnetic resonance imaging is widely used in clinical diagnostic radiology, and magnetic nanoparticles have been employed in the manipulation of surface patterns, cell sorting, mechano-conditioning of cells, and cellular micromanipulation, among other applications.
Under the conditions used for creating levitated 3-D cell culture, magnetic iron oxide nanoparticles are well tolerated by mammalian cells, a result consistent with previous reports.
To form levitated 3-D cultures, Dr. Killian and his coworkers incubate the cells with the gel, allowing the cells to absorb the magnetic nanoparticles. Excess gel is then washed away. Application of an external magnet causes the treated cells to rise to the air-medium interface, and within 12 hours multicellular structures assemble themselves.
In experiments with neural stem cells, levitated structures formed branching configurations consistent with aggregated cell clusters. Human glioblastoma cells displayed certain biochemical functions (N-cadheren production) similar to in vivo tissue.
Finally, Dr. Killian argued that magnetic field manipulation may provide new opportunities for culturing and manipulating mixed populations of cells. When human glioblastoma cells and normal human astrocytes were separately cultured and then guided together by external magnetic fields, a clear interface separating the cell structures was initially evident.
By 12 hours, the populations began to fuse, lose their individual spherical shapes, and coalesce into a single spheroid with the human glioblastoma cells invading the structure composed of normal human astrocytes.
“These results illustrate the potential of this methodology for analysis of brain tumor invasiveness in co-culture assays, suggesting that 3-D culturing through magnetic levitation of cells is a useful biotechnology,” Dr. Killian concluded.
Nano3D Biosciences’ Bio-Assembler™ system cultures cells in three dimensions by magnetic levitation. The Bio-Assembler uses nanoparticle-based Nanoshuttle-PL solution to deliver magnetic nanoparticles to cells. Magnetic drives then levitate cells to create the 3-D cell growth environment.



