Stephen Lappin
Mitu Chaudhary
Certain consideration can help laboratories to derive the greatest return from their automation investments.
Automating sample preparation for next-generation sequencing (NGS) offers advantages beyond increased throughput, such as improved data consistency. Unlike manual methods where variability increases with an increased sample set, an automated system provides consistent data quality whether you are processing 1 or 1,000 reactions.
A variety of automation choices are available in the market today that increase throughput, provide consistency in results, streamline workflow, and potentially provide walkaway time to researchers so that they can perform valuable tasks.
In the previous article, we considered how to ease the transition from manual to automated NGS sample preparation. In this article, however, we go one step further. We consider how to sustain the benefits of automation. The following considerations determine the success of automation in your laboratory.
Using Compatible Labware
This is a very important consideration in automation. A similar-looking plate from a different vendor may not be an appropriate alternative to a recommended/validated plate. Plates from different vendors, with different material and finish, can affect the protocol, and thus the resulting data.
Some might argue that the pipette tips are the most important piece of labware in the process. As with manual methods, DNA/RNA free and filter tips from a quality supplier are strongly recommended for automated systems to protect precious samples.
Minimizing Reagent Waste
Batching of samples can minimize waste when automating the sample preparation protocol. That’s because automation protocols require more dead volume than the same process done manually. Many standard kits do not have enough dead volume to provide for the number of samples quoted with the kit. This can add to the cost per sample.
At least some part of the increase in reagent volume can be offset over time by eliminating the wastage due to manual errors. Kits that are developed specifically for automation usually have the extra reagent volume needed. Regardless, when working with automated systems, a minimum batch size of 24 reactions will give the greatest efficiency in terms of reagent use.
Accommodating Users of Different Skill Levels
Tiered access for power users and end users can benefit laboratories that intend to manage user permissions with protocols. For example, a two-tiered schema could enable power users to create new protocols or modify existing ones while allowing technicians to run predetermined protocols. These protocols could be accessed via on-screen instructions, which could lead technicians through system set up and other tasks (Figure).
See this article’s prequel for lab set up considerations. Also, be sure to check out the infographic “NGS Sample Prep—Manual vs. Automated“.
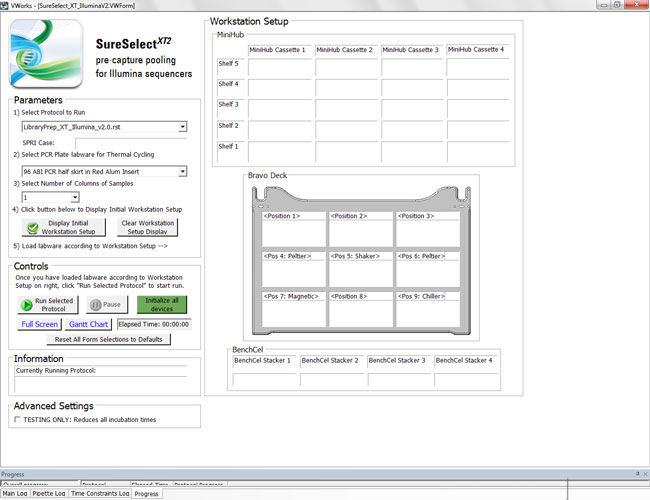
Figure. Form feature in Agilent Automation protocols enable a routine user to run protocols without learning programming.
Stephen Lappin is applications scientist, and Mitu Chaudhary ([email protected]) is product manager at Agilent Technologies. More information on Agilent NGS Sample Prep Automation.







